Transcriptional activation of genes within the nucleus of eukaryotic cells occurs by a variety of mechanisms. Typically, these mechanisms rely on the interaction of regulatory proteins (transcriptional activators or repressors) with specific DNA sequences that control gene expression. Upon DNA binding, regulatory proteins also interact with other proteins that are part of the RNA polymerase II transcriptional complex.
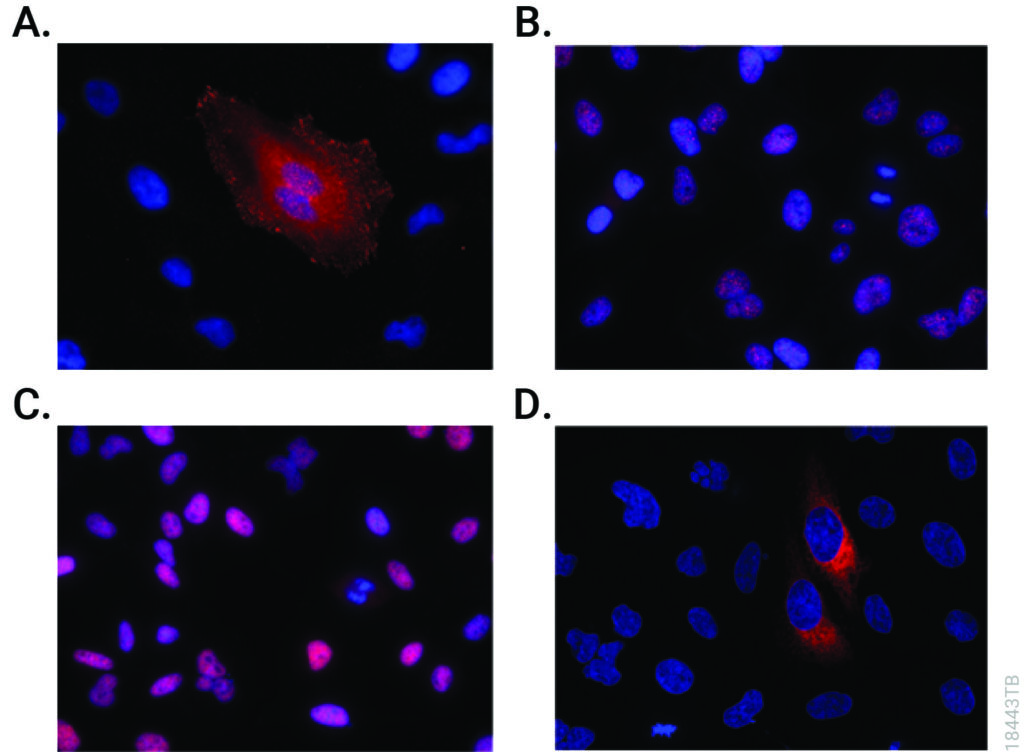
One type of transcriptional activation relies on inducing a conformational change in chromatin, the DNA-protein complex that makes up each chromosome within a cell. In a broad sense, “extended” or loosely wound chromatin is more accessible to transcription factors and can signify an actively transcribed gene. In contrast, “condensed” chromatin hinders access to transcription factors and is characteristic of a transcriptionally inactive state. Acetylation of lysine residues in histones—the primary constituents of the chromatin backbone—results in opening up the chromatin and consequent gene activation. Disruption of histone acetylation pathways is implicated in many types of cancer (1).
Continue reading “Designing BET(ter) Inhibitors to Guide Therapy for Cancer and Inflammatory Diseases”