For the success of adeno-associated virus (AAV)-based gene therapies, accurate viral titration is non-negotiable. But as interest in AAVs as delivery vectors soars, so does the challenge of getting consistent, reproducible genome titers—a critical hurdle in biologics workflows where speed and standardization are paramount.
Impact of DNase Digestion on Accuracy
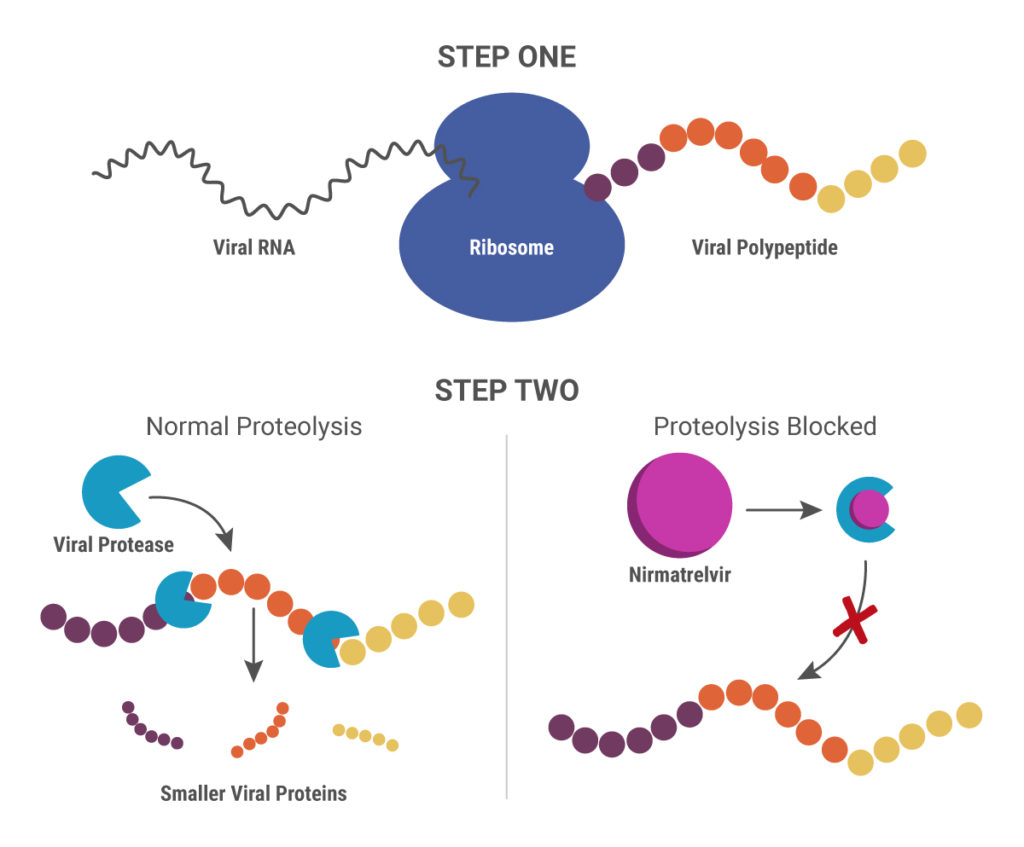
A recent peer-reviewed study pinpoints a surprising source of this variability: the DNase digestion, a common step used to remove contaminating DNA. “[DNase digestion]… led to a significant decrease in genome titers for several AAV serotypes,” the authors write,highlighting concerns around workflow reproducibility and data reliability.The research, published in Molecular Therapy: Methods & Clinical Development, demonstrates how different engineered AAV serotypes respond inconsistently to standard DNase treatment, significantly impacting final titer results. These findings are particularly relevant for scientists developing and optimizing cell and gene therapy platforms, where regulatory expectations for analytical precision continue to rise.
This study addresses the challenge of accurately measuring viral titers in engineered AAVs, which have enhanced transduction efficiency but exhibit lower yields when measured using traditional genome titering methods. Specifically, the authors explored the impact of DNase digestion on the stability of engineered AAV capsids that contain peptide insertions. Through a series of rigorous experiments including electron microscopy, quantitative PCR (qPCR) and digital droplet PCR (ddPCR), they found that the heat-inactivation step commonly used following DNase treatment to eliminate free-floating DNA can compromise the integrity of engineered AAV capsids.
Continue reading “Solving the AAV Titer Challenge: A New Approach to Gene Therapy Workflows “