Today’s blog is written by guest blogger Kristin Huwiler from our Cellular Analysis and Proteomics Group.
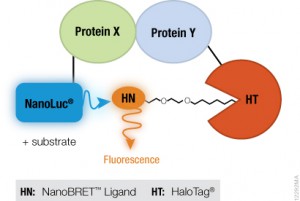
Two research collaborations, one in Europe and a second in the US, have just published in Nature Chemical Biology (1,2) on the identification of BET inhibitors (bi-BETs) that bind via a bivalent mechanism to both bromodomains of BRD4. These bivalent chemical inhibitors exhibit high cellular potency and affinity relative to their monovalent predecessors. By developing high-affinity ligands that engage both bromodomains simultaneously within BRD4, the authors illustrate a concept that may be applicable in the development of selective, potent ligands for other multi-domain proteins. Here we review the work presented in the Waring et al. paper using the Promega NanoBRET™ Technologies to characterize the mechanism of action of their bivalent probe.
The bromodomain and extraterminal (BET) sub-family are some of the most studied bromodomain-containing proteins (3). The BET subfamily of proteins contain two separate bromodomains. BRD4 is one well studied member of the BET sub-family. Several small molecule inhibitors that target BRD4 have been developed as potential therapeutics for various cancers with promising initial studies, but to date are all monovalent, binding each bromodomain of the BET family members separately (2).