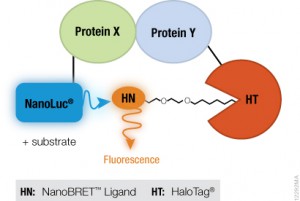
Genetic reporters are used as indicators to study gene expression and cellular events coupled to gene expression. They are widely used in pharmaceutical and biomedical research and also in molecular biology and biochemistry. Typically, a reporter gene is cloned with a DNA sequence of interest into an expression vector that is then transferred into cells. Following transfer, the cells are assayed for the presence of the reporter by directly measuring the reporter protein itself or the enzymatic activity of the reporter protein. A good reporter gene can be identified easily and measured quantitatively when it is expressed (in the organism or cells of interest).
Genetic reporters are used as indicators to study gene expression and cellular events coupled to gene expression. They are widely used in pharmaceutical and biomedical research and also in molecular biology and biochemistry. Typically, a reporter gene is cloned with a DNA sequence of interest into an expression vector that is then transferred into cells. Following transfer, the cells are assayed for the presence of the reporter by directly measuring the reporter protein itself or the enzymatic activity of the reporter protein. A good reporter gene can be identified easily and measured quantitatively when it is expressed (in the organism or cells of interest).
Bioluminescent reporters are ideal for these types of studies because they have a number of important features including:
• Measurements that are almost instantaneous
• Exceptional sensitivity
• A wide dynamic range
• Typically no endogenous activity in host cells to interfere with quantitation
However, one factor that is critical for the success of a bioluminescent reporter assay is the vector.
At Promega we offer several different luciferases as reporters, and the genes for those luciferases are available in a variety of vectors. The vectors may vary in the promoters used or the presence or absence of sequences for rapid degradation. Often seemingly small changes in the vector can make a big difference in the suitability of the vector for a given experimental system. Do you need a reporter with a short half-life to detect rapid changes in gene expression? Are you studying a specifically localized protein? Do you wish to perform a transient or stable transfection?
To make finding the best reporter vector for your experimental system easy, we have developed the Luciferase Reporter Vector Selector. Using this online tool, you can narrow the choices of available vectors by promoter type, application (in vivo imaging, cancer pathway analysis, etc), availability of selectable marker, and type of luciferase.
So, as you design your luciferase reporter experiment, keep in mind this handy tool to help you choose the best luciferase vector for your needs.


 Timing is everything! I learned that the hard way just two weeks ago when I took my son to scout camp and thought I would try to capture the traditional American flag ceremony for posterity. I set up my camera for a panoramic shot and scanned the crowd. Feeling very pleased with myself, I got home that evening ready to show my family the great camera skills I had honed over the Summer months. To my horror, I noticed that half of the scout troop was saluting the flag while the other half were standing to attention! I had got the timing horribly wrong (although the picture is still fun to look at in a strange sort of way).
Timing is everything! I learned that the hard way just two weeks ago when I took my son to scout camp and thought I would try to capture the traditional American flag ceremony for posterity. I set up my camera for a panoramic shot and scanned the crowd. Feeling very pleased with myself, I got home that evening ready to show my family the great camera skills I had honed over the Summer months. To my horror, I noticed that half of the scout troop was saluting the flag while the other half were standing to attention! I had got the timing horribly wrong (although the picture is still fun to look at in a strange sort of way).