The path to drug development is strewn with obstacles: Identifying targets; configuring assays to help identify targets or drugs; uncovering the right compound to affect the selected target without off-target effects and screening multiple compounds to eliminate or identify potential drugs. Without the right tools, compounds or target, identifying potential disease therapies becomes nearly impossible.
When it comes to a drug target for cancer, the Ras protein family is at the top of the list because the proteins are expressed ubiquitiously and found mutated in many types of cancer. Because Ras proteins are involved in transducing signals from the surface of cells, many of the resulting mutations produce an activated Ras, inducing uncontrolled expression of the genes that Ras controls. Ras proteins are small GTPases (20–25kDa) that comprise a larger superfamily of proteins divided into five subfamilies: Ras, Rho, Rab, Arf, and Ran. These proteins control diverse cellular activities, including cellular differentiation, proliferation, cell division, nuclear import and export, and vesicle transport. GTPases are guanosine-nucleotide-binding proteins with affinity for GDP or GTP and are able to hydrolyze GTP. When bound to GTP, GTPases are active (turned on) and interact with downstream proteins in the signaling cascade. When GTPases are bound to GDP, the proteins are inactivated (turned off) and no longer transduce signals.
Continue reading “A Better GTPase Assay for Drug Development”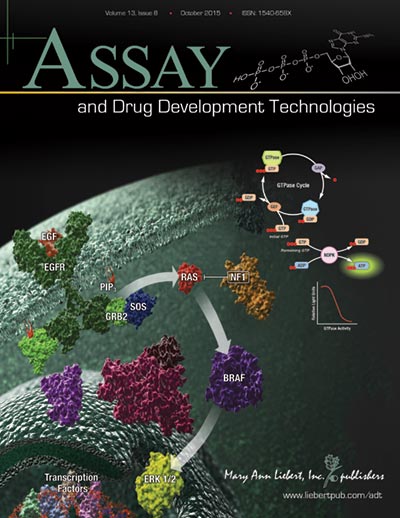


 Antibodies labeled with small molecules such as fluorophore, biotin or drugs play a critical role in various areas of biological research,drug discovery and diagnostics. There are several limitations to current methods for labeling antibodies including the need for purified antibodies at high concentrations and multiple buffer exchange steps.
Antibodies labeled with small molecules such as fluorophore, biotin or drugs play a critical role in various areas of biological research,drug discovery and diagnostics. There are several limitations to current methods for labeling antibodies including the need for purified antibodies at high concentrations and multiple buffer exchange steps. Often a diagnosis of thyroid cancer is associated with a good prognosis and fairly straightforward surgical treatments to remove the tumor followed by radioactive iodine ablation. Such treatment works well in tumors that have not metastasized and retain enough of their thyroid cell “identity” that they can still accumulate radioactive iodine.
Often a diagnosis of thyroid cancer is associated with a good prognosis and fairly straightforward surgical treatments to remove the tumor followed by radioactive iodine ablation. Such treatment works well in tumors that have not metastasized and retain enough of their thyroid cell “identity” that they can still accumulate radioactive iodine.