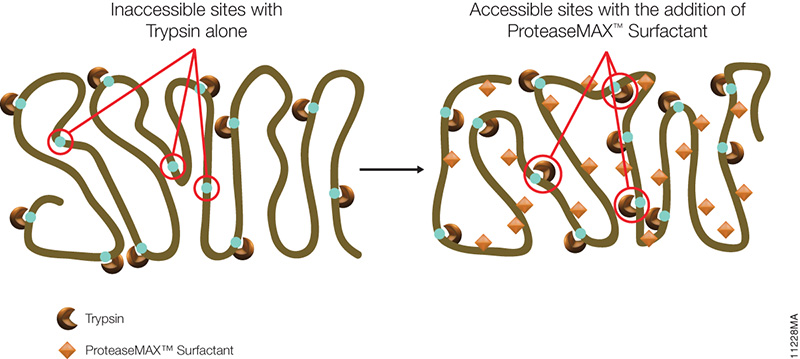
The novel mass spectrometry compatible surfactant sulfonate-(sodium 3-((1-(furan-2-yl)undecyloxy) carbonylamino)-propane-1-sulfonate (i.e.ProteaseMAX) facilitates both in-gel and in-solution digestion applications by reducing the time required, enabling protein solubilization/denaturation and increasing peptide/protein identifications.
A new application was highlighted in a recent publication (1) which utilized ProteaseMAX to lyse cells prior to trypsin digestion and subsequent mass spec analysis. The composition of the buffer determines the overall efficiency of cell lysis, dissociation of protein complexes, protein solubility and ease of removal prior to LC/MS-MS analysis.
Are you looking for proteases to use in your research?
Explore our portfolio of proteases today.
When compared to lysis buffers containing either urea or SDC, ProteaseMAX provided the optimal number of identified peptides/proteins.
In addition it can be easily removed from the lysate by acidic precipitation.
Reference
- Pirmoradian, M. et al. (2013). Rapid and deep human proteome analysis by single-dimension shotgun proteomics. Mol. Cell. Prot. 12, 3330–8.

test comment