
Mass spectrometry depends on the successful digestion of proteins using proteases. Many commercially available proteomic-grade trypsins contain natural contaminants that produce non-specific cleavages. Trypsin Platinum, a new protease from Promega provides maximum specificity, giving you cleaner and more conclusive data from mass spec.
Trypsin is typically extracted from bovine or porcine pancreas. In addition to trypsin, both of these sources also contain chymotrypsin. To suppress chymotryptic activity, trypsin is treated with tosyl phenylalanyl chloromethyl ketone, or TPCK, to irreversibly inhibit the chymotrypsin. However, trace amounts of chymotrypsin appear to escape this inhibition and produce non-specific cleavages, as seen in the figure below.
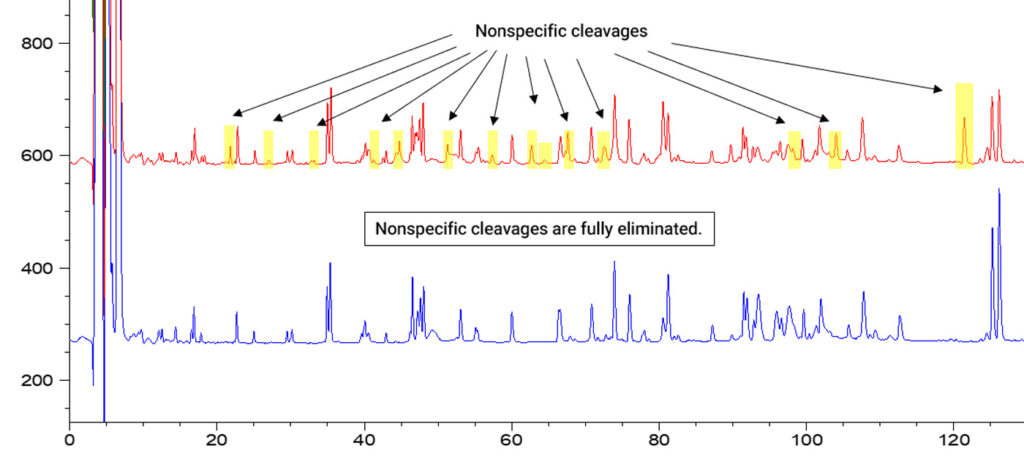
Autoproteolysis can also complicate trypsin digestion. Protein mass spec-grade trypsins are chemically modified to suppress autoproteolysis, but certain levels can still be observed.
Trypsin Platinum, Mass Spectrometry Grade, is free of any detectable nonspecific proteolytic activity. A novel chemical modification assures maximal autoproteolytic resistance. This results in the most optimal trypsin available for users looking for accurate protein characterization with mass spectrometry.
Try a sample of high-efficiency Trypsin Platinum today!
Watch our on-demand webinar or visit our website for more on Trypsin Platinum, Mass Spectrometry Grade, with enhanced proteolytic efficiency and superior autoproteolytic resistance.
Want to see how Promega mass spec reagents are being used in recent research?
Check out these blog posts:
- A New Method to Detect Cholangiocarcinoma Biomarkers – Promega Mass Spec Grade Trypsin/Lys-C Mix was used to identify and quantitate proteins present in extracellular vesicles.
- Mass Spec for Glycosylation Analysis of SARS-CoV-2 Proteins Implicated in Host-Cell Entry – Trypsin. Chymotrypsin, Asp-N and Lys-C were used to digest the spike protein of SARS-CoV-2.
Are you looking for proteases to use in your research?
Explore our portfolio of proteases today.
