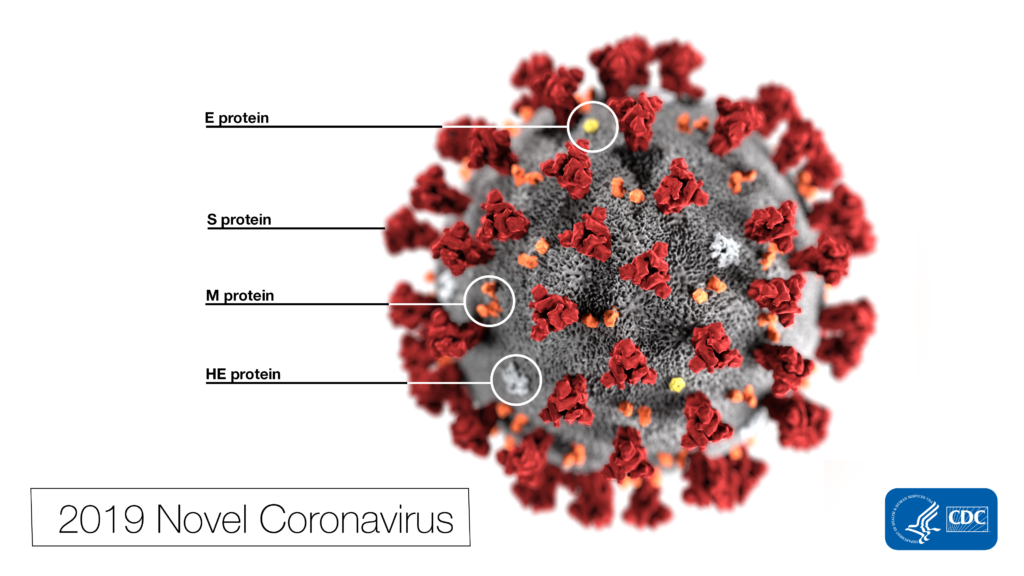
As the SARS‐CoV‐2 pandemic continues to rage across the United States and around the globe, the demand for COVID‐19 testing is increasing. The vast majority of the COVID-19 assays use RT‐qPCR to detect the viral RNA in patient samples such as nasopharyngeal swabs, which are collected and stored in viral or universal transport media (VTM/UTM). The general workflow for these COVID‐19 assays can be broken down as follows:
- Collect and store patient samples
- Ship samples to testing laboratory
- Extract RNA from samples
- Amplify and analyze samples
While many companies who manufacture the products that are used in these steps have been able to adapt and significantly increase their production capacities, there are still gaps between the supply of these products and the global test demand. Both the sample collection and storage step and the RNA extraction/purification step have a tendency to bottleneck and experience supply constraints. One way to address these bottlenecks and expand production capacity for these in‐demand products is to evaluate the viability of skipping a step in the workflow, without hindering the ability to detect viral RNA from samples.
Continue reading “XpressAmp™ Direct Amplification: Simplified and Accelerated Time to qPCR Results”