Imagine you are traveling in your car and must pass through a mountain range to get to your destination. You’ve been following a set of directions when you realize you have a decision to make. Will you stay on your current route, which is many miles shorter but contains a long tunnel that cuts straight through the mountains and obstructs your view? Or will you switch to a longer, more scenic route that bypasses the tunnel ahead and gets you to your destination a bit later than you wanted?

Choosing which route to take illustrates a clear trade-off that has to be considered—which is more valuable, speed or understanding? Yes, the tunnel gets you from one place to another faster. But what are you missing as a result? Is it worth a little extra time to see the majestic landscape that you are passing through?
Considering this trade-off is especially critical for researchers working with human DNA purified from formalin-fixed paraffin-embedded (FFPE) or circulating cell-free DNA (ccfDNA) samples for next-generation sequencing (NGS). These sample types present a few challenges when performing NGS. FFPE samples are prone to degradation, while ccfDNA samples are susceptible to gDNA contamination, and both offer a very limited amount of starting material to work with.
If degradation or contamination are not detected before NGS library preparation and sequencing, the sequencing run will give limited results due to a lack of amplifiability. Not only does this mean additional time and cost to repeat the study, but samples crucial to an investigation may have been wasted if there is not enough sample remaining to start over.
Luckily, amplifiability is something that can be assessed to ensure that samples are not wasted. For example, you can use the ProNex® DNA QC Assay to determine the quality, quantity and amplifiability of the DNA you purified from your sample. Samples with a 75/300bp ratio unacceptable relative to your lab’s cutoff point would not be considered amplifiable and would not be prepared for sequencing at all or until further cleanup steps had been completed.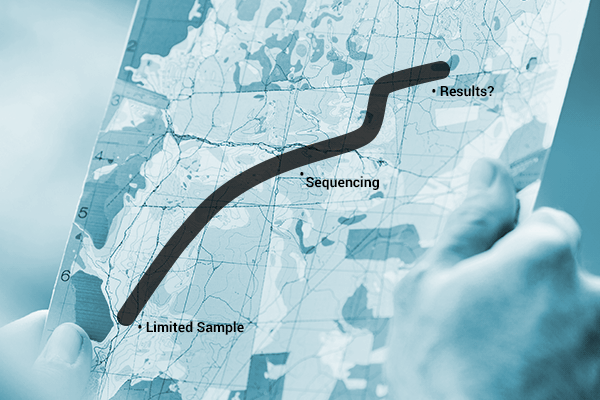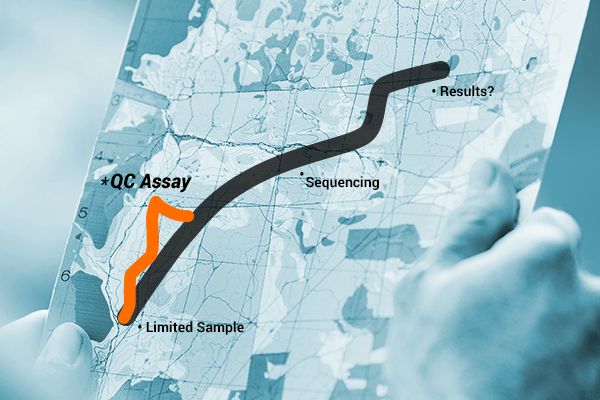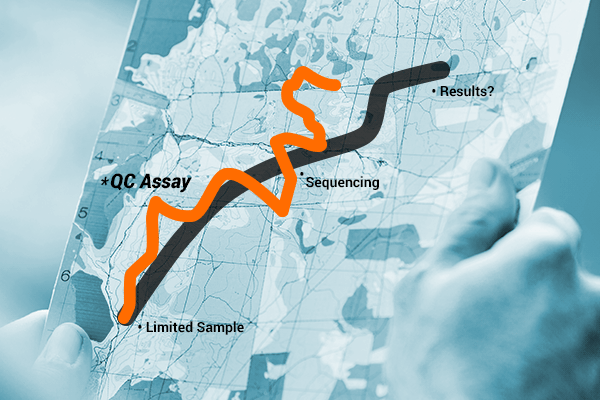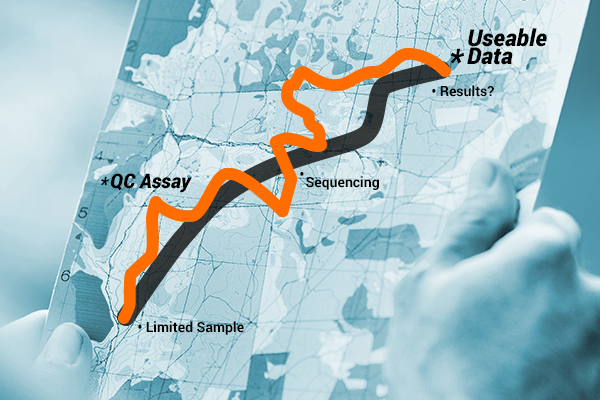
Think about your NGS workflow like the trip through the mountains I mentioned earlier, where you are presented with multiple routes. You could choose a workflow with fewer steps (the shorter route) which may or may not require backtracking to perform additional steps or result in null results. Like entering a tunnel, this path prevents you from taking a closer look at your sample which could mean you don’t get usable data.
While you can save time and effort by immediately sequencing your purified DNA, there is a real risk that you may not get the desired results because your DNA is not amplifiable. Taking a bit of extra time to seek out more information about your samples is small price to pay in order to gain confidence that you will not be wasting precious samples and will get the results you need.
Alternatively, you could choose to include an extra step in your workflow and QC your samples to assess for amplifiability before sequencing (the longer, less direct route). This route gives you a full view of your sample, and the extra time it takes allows you to gather the information necessary to determine if you can proceed with sequencing and expect good data. Ultimately, the additional time and effort eliminates the risk of wasting samples or repeating workflow steps.
When performing NGS with FFPE and ccfDNA samples, sequencing without verifying quality, quantity and amplifiability is not worth the risk. You really do need to see the whole picture and QC your samples, even if it does take an extra step. So, choose the scenic route, sit back and enjoy getting to know your DNA.
Latest posts by Darcia Schweitzer (see all)
- Solving the AAV Titer Challenge: A New Approach to Gene Therapy Workflows - December 10, 2025
- Cytochrome P450 Inhibition: Old Drug, New Tricks - May 5, 2022
- Firefly Luciferase Sheds Light on Development of New Malaria Treatments - April 5, 2021
