Our skin, respiratory system and gastrointestinal tract are continually bombarded by environmental challenges from potential pathogens like SARS-CoV-2. Yet, these exposures do not often cause illness because our immune system protects us. The human immune system is complex. It has both rapid, non-specific responses to injury and disease as well as long-term, pathogen-specific responses. Understanding how the immune response works helps us understand how some pathogens get past it and how to stop that from happening. It also provides key information to help us develop safe and effective vaccines.
The immune response involves two complementary pathways: Innate Immunity and Adaptive Immunity. Innate immunity is non-specific, rapid and occurs quickly after an injury or infection. As a result of the innate immune response, cytokines (small signaling molecules) are secreted to recruit immune cells to an injury or infection site. Innate immunity does not develop “memory” of an antigen or confer long-term immunity.
The immune response involves to complementary pathways: Innate Immunity and Adaptive Immunity.
Unlike innate immunity, adaptive immunity is both antigen-dependent and antigen-specific, meaning that adaptive immune response requires the presence of a triggering antigen—something like a spike protein on the surface of a virus. The adaptive immune response is also specific to the antigen that triggers the response. The adaptive immune response takes longer to develop, but it has the capacity for memory in the form of memory B and T cells. This memory is what enables a fast, specific immune response (immunity) upon subsequent exposure to the antigen.
Adaptive and innate immunity work in concert when the body is exposed to pathogens, including viruses or other injury. Dysregulation or defects in either pathway can result in illness or diseases such as autoimmune diseases, immunodeficiencies, hypersensitivity reactions and even the cytokine storm phenomenon associated with certain viral infections. The remainder of this article will focus solely on the adaptive immune response.
Adaptive Immune Response
The main role of the adaptive immune response is to differentiate “self” from “non-self” and once “non-self” antigens are recognized, generate a response that targets the host cells or pathogens bearing the “non-self” antigen for destruction.
The adaptive immune response can be further subdivided into two response pathways: cell-mediated and humoral (antibody), although the two pathways “intersect” and feedback on each other. The cells involved in the adaptive immune response include T cells and B Cells.
T Cell Activation in the Adaptive Immune Response
T cells can be divided into two major classes based on the receptors they express on their cell surface. All T cells express the T-cell Receptor (TCR), which is unique to the cell and recognizes specific antigens presented by the Major Histocompatibility Complex (MHC 1 or MHC II) on “antigen presenting cells” (APC). [APCs are host cells that process proteins and present the fragments on their cell surface.]
“T helper cells” (Th Cells) are T cells that express the CD4 protein (CD4+) and are activated by MHC II presenting APCs. Cytotoxic T cells are T cells that express the CD8+ protein and are activated by MHC I-presenting APCs. T cells are activated from their immature or naïve state (never bound to antigen) to an active or effector state once they bind antigen and receive several other signals from the APC.
Cytotoxic T cells (CD8+) are involved in the destruction of infected host cells that present the recognized antigenic peptides on their cell surfaces (e.g. pieces of a viral spike protein). Cytotoxic T-cells proliferate by clonal expansion in response to the infection. Once the infection is cleared, most cytotoxic T cells die; however, a few will remain circulating as memory T cells that become activated again if they encounter the same antigen.
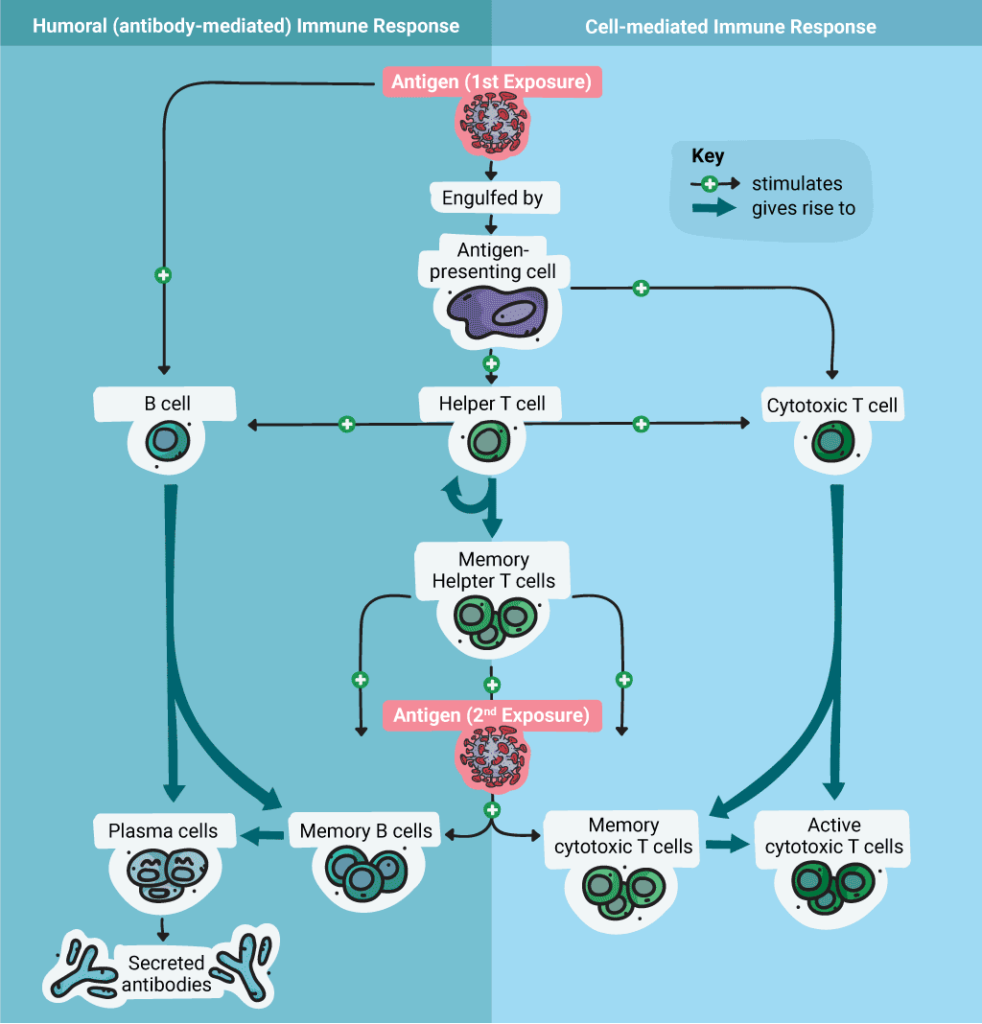
T Helper Cells mediate the immune response by directing other cells (e.g., macrophages) to kill infected host cells and clear pathogens. They are activated by binding to a specific antigen presented on an MHC II complex by an APC. When Th cells are activated, they release cytokines that further mediate and stimulate immune response.
B Cell Activation and Antibody Production in the Adaptive Immune Response
The antibody response, also known as the humoral immune response, results from the activation of B cells. B cells are produced in the bone marrow, and they each express a unique antigen-binding receptor on their membranes. They can recognize an antigen directly and don’t require an antigen presenting cell. The main function of B cells is to produce antibodies to a specific antigen.
When a B cell is activated by an antigen, the B cell proliferates and produces two types of cells: antibody-secreting plasma cells and memory B cells. Memory B cells are long-lived cells that express antigen-specific receptors so that the antigen can be recognized quickly upon a second exposure. Plasma cells secrete antibodies specific to the target “non-self” antigen.
B cell activation begins when the B cell antigen binding receptors bind the antigen. When this happens, Th cells are attracted, and they secrete cytokines that promote B cell proliferation and differentiation into Memory and Plasma cells. The plasma cells secrete antibodies that bind to the antigen.. These antibodies promote processes including complement activation (which enhances the innate immune response, targeting pathogens or infected cells for destruction), opsonization and phagocytosis and elimination of the pathogen.
Antibodies
Antibodies are important for preventing virus proliferation during the initial (acute) phases of infection, but once a viral infection is established, the cell-mediated response of cytotoxic T-cells, natural killer cells and macrophages that targets infected host cells for destruction and stimulates additional cytokine production.
There are five kinds of human immunoglobulins (Ig) antibodies produced during an immune response. IgM is the first Ig produced during B cell development. It triggers opsonization of a pathogen and complement cascade. IgG is usually produced during a secondary immune response. IgG molecules are important for neutralizing toxins or viruses, targeting pathogens for destruction via opsonization, and complement fixation. IgG are also the only kind of immunoglobulin that can cross the placenta. IgA molecules are involved in protecting mucosal surfaces from pathogens. IgE molecules are important for responses to parasites and play an key role in hypersensitivity and allergies. Finaly, the function of the IgD molecule is not completely understood.
There are five kinds of immunoglobulins (Ig) antibodies produced during an immune response.
Many serological assays that test for the presence of specific antibodies as evidence of exposure to a pathogen detect IgM and/or IgG antibodies. Such tests can be used in the clinic to determine if a person has been exposed to a pathogen by detecting if they have mounted an immune response (i.e., produced antibodies). However, the presence of antibodies does not always mean that the person has developed long-term immunity. And for some viruses, a successful viral response may be more dependent on the cell-mediated immunity of T cells than humoral immunity.
Applying What We Know about Immunity to Vaccine Development
Serological tests are also key tools for researchers who are attempting to design and produce effective vaccines. Creating a vaccine that generates long-term effective immunity against a disease-causing pathogen is challenging. Not only do the vaccines need to generate an immune response in the vaccinated individual that results in the production of significant antibodies, but those antibodies need to possess certain qualities including specificity, neutralizing ability, and strong binding to the antigen. Additionally the immune response to the vaccine needs to be sufficient such that memory is generated through the B cell and T cell pathways and persists in the vaccinated individual for a long time. Developing vaccines to respiratory viruses pose additional challenges in that we want to be sure that the vaccine does not result in undesirable effects such as damaging inflammation in the lungs. Understanding how a pathogen enters a host, triggers the immune response, and even corrupts the immune response to its own advantage is key to developing safe and effective vaccines.
Further Reading and Sources on Basic Immunology
Warrington, R. et al. (2011) An Introduction to Immunology and Immunopathology. Allergy, Asthma and Clinical Immunology Vol 7, Supplement 1.
Janeway, C.A. et al. (2001) Immunobiology, 5th ed. The Immune System in Health and Disease. Garland Science, New York.
The College of Physicians of Philadelphia. The History of Vaccines: How Vaccines Work [Internet: https://www.historyofvaccines.org/content/how-vaccines-work Accessed -7/2/2020]
The College of Physicians of Philadelphia. The Human Immune System and Infectious Disease [Internet: https://www.historyofvaccines.org/content/articles/human-immune-system-and-infectious-disease Accessed -7/2/2020]
