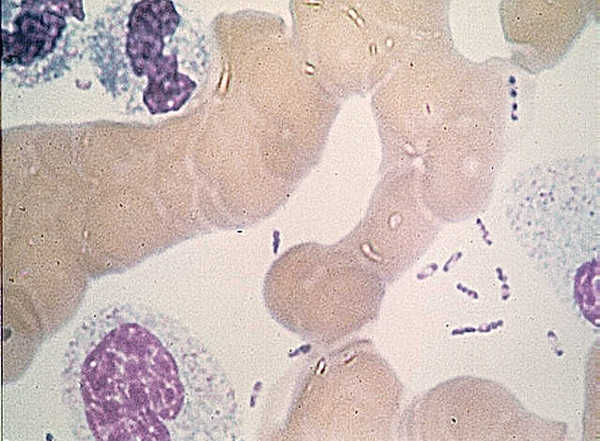
Gonzales et al. gave intradermal injections to female C57BL/6J mice using 200 CFU of Y. pestis strain CO92, 2µl total volume, with both the volume and the CFU mimicking a flea bite to mouse ear. Y. pestis was tagged with green fluorescent protein (gfp) or red fluorescent protein (rfp). Whole mounts of mouse ear were then examined by confocal microscopy. By using the whole organ, researchers were minimally disturbing tissues architecture while tracking Y. pestis dissemination. Bacteria were found in two places in the mouse ear: The injection site and the base of the ear in tube-like structures presumed to be lymphatic vessels. While the presence of Y. pestis at the base of ear occurred in 10% of the mice at 24 hours post inoculation (hpi), the researchers found it significant due to the strong signal from this area even at low magnification. Staining with DAPI, a fluorescent dye that binds to DNA, showed host cells in close proximity to the bacteria present in the presumptive lymphatic vessels.
To detect if Y. pestis was present in the LN and lymphatic vessels that connect the ear with lymph nodes, the infected ear area and adjacent tissue that included the draining lymph node and lymphatic vessels that connect both tissues. Twenty-four hours post inoculation, the fluorescent-protein-expressing bacteria were present in the afferent lymphatic vessels, those that drain into the lymph node. At 48hpi, these areas had even stronger fluorescent signals. In both 24 and 48hpi, DAPI staining revealed the presence of host cells associated with the bacteria.
Based on the hypothesis that the majority of the bacteria on the skin find their way to the lymph node, Gonzales et al. developed 10 oligonucleotide-tagged Y. pestis strains. These oligo tags did not affect bacterial infectivity and could be used to follow the course of infection from inoculation site to lymph node. Mice were inoculated with a mixture of nine tagged strains (the tenth strain was used as a control) and the progress of infection assessed by Southern dot blot. At 12hpi, researchers found an average of 2.2 strains (range 1–7) present in the lymph node. At 48hpi, there were an average of 2.8 strains (range 1–5) found in the lymph node. When spleens, an organ the reflects the systemic dissemination of Y. pestis, were removed and tested for the tagged strains, only the tagged strains that were present in the lymph node were found in the spleen. Based on these observations, the researchers suggested that there was a bottleneck getting from the skin to the lymph node, but once a strain made it to the lymph node, it spread to cause infection in the mouse. One mouse was an exception to this observation: There were five strains in the lymph node and only two spread systemically. Taken together, these results indicate that once the strains make it to the lymph nodes, they are the only populations from which the systemic infection originates.
What happens to this bottleneck if the bacterial inoculum is increased tenfold to 2,000CFU? Of the eight mice tested under this increased infection burden, only four had all nine tagged strains; the others had six, seven or eight strains in the lymph node. While not as reduced as the 200CFU injection (average strain in lymph node = 3), this outcome suggested that a population bottleneck still occurred and prevented all of the strains from making it to the lymph node for dissemination.
Is this bottleneck phenomenon limited to the ear? When Gonzales et al. compared mice inoculated by ear or foot, the same limitation of strains occurred. That is, a median of three strains made it the lymph node and a median of two were found in the spleen for those inoculated on the foot. Researchers also bypassed the dermis to subcutaneously infect the mice, a way to test if the population selection only occurs on the skin. In fact, half of the mice infected this way had all nine strains present in the lymph node with a median of eight strains for all the mice tested. Therefore the strain selection seems related to the skin and bypassing the skin eliminated this population bottleneck.
Does something in the skin kill the bacteria before it can make it to the lymph node? If this is true, the number of strains on the skin and in the lymph node should be equal. However, examining the skin surface 12 and 48hpi showed a greater number of strains (four to eight) on the skin versus average of three in lymph node. To test if the skin was altered in response to the presence of Y. pestis, mice were inoculated with a carbenicillin-resistant strain and 24hpi the same animals inoculated in the same location with a kanamycin-resistant strain. Forty-eight hours after the initial inoculation, bacteria from lymph nodes were plated on medium containing either carbenicillin or kanamycin and the bacterial load compared to control mice injected with PBS at the first inoculation. The researchers found fewer bacteria resistant to kanamycin from the lymph nodes of mice whose ears were previously exposed to bacteria compared to control mice inoculated with PBS. Gonzales et al. suggest that interaction of the skin with Y. pestis activates the tissue, affecting the ability of the bacteria to disseminate to the lymph node and those bacteria that do make it to the lymph nodes happen prior to this skin activation.
Based on other work, neutrophils play a role in Y. pestis infection on skin so the researchers focused on these immune cells. Using the fluorescently labeled α-Ly6G, an antibody that binds to neutrophils, the proximity of bacterial cells and neutrophils were tracked by confocal microscopy. Starting at 4hpi, many bacteria were in close proximity to neutrophils or inside them, and at 24hpi, there seemed to be a sharp increase in the number of observed neutrophils but few bacterial cells were associated at this later time. The neutrophils were noted as highly concentrated at the injection site. Gonzales et al. considered that neutrophils may be involved in bacterial movement to the lymph nodes but depleting the neutrophils did not change the population bottleneck in the lymph nodes. However, when the same neutrophil-depleting antibody was applied to the skin, the bacteria burden increased compared to mice treated with PBS. Thus, neutrophils play a role in reducing bacterial proliferation on the skin and affecting which strains reach the lymph nodes.
The research in this PLOS Pathogens paper demonstrated that there is a Y. pestis population bottleneck from simulated flea bite infections as the bacteria moves from the skin to the lymph node. Furthermore, neutrophils on the skin surface seem to be the mechanism for this strain selection once they respond to the presence of bacteria. This information increases our understanding of infection by a bacterial pathogen and offers another mechanism for studying an in vivo model of infection.
Reference
Gonzalez R.J., Lane, M.C., Wagner, N.J., Weening, E.H. and Miller, V.L. (2015). Dissemination of a Highly Virulent Pathogen: Tracking The Early Events That Define Infection, PLOS Pathogens, 11 (1) e1004587. DOI: http://dx.doi.org/10.1371/journal.ppat.1004587
Sara Klink
Latest posts by Sara Klink (see all)
- A One-Two Punch to Knock Out HIV - September 28, 2021
- Toxicity Studies in Organoid Models: Developing an Alternative to Animal Testing - June 10, 2021
- Herd Immunity: What the Flock Are You Talking About? - May 10, 2021
