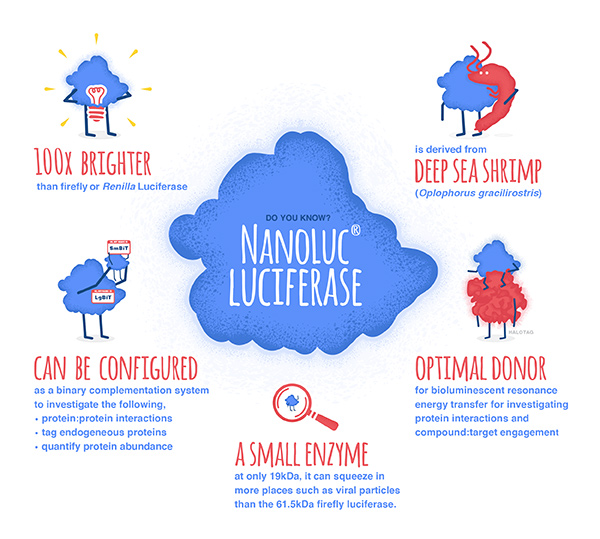
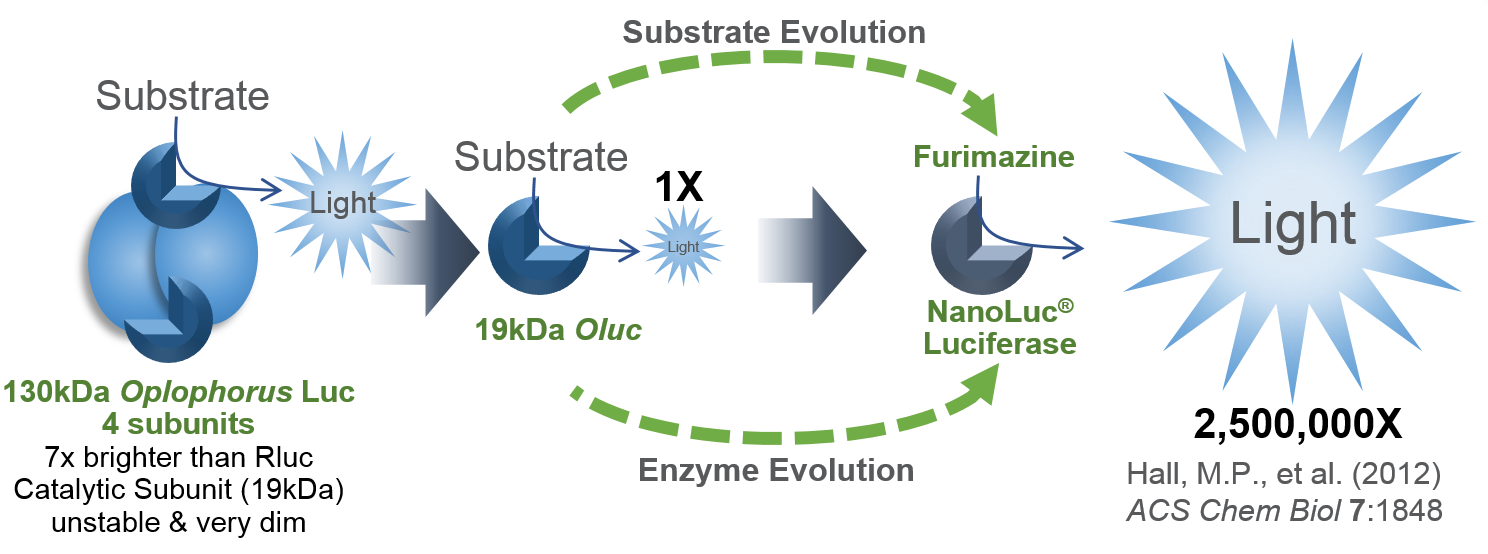
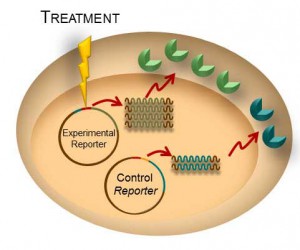
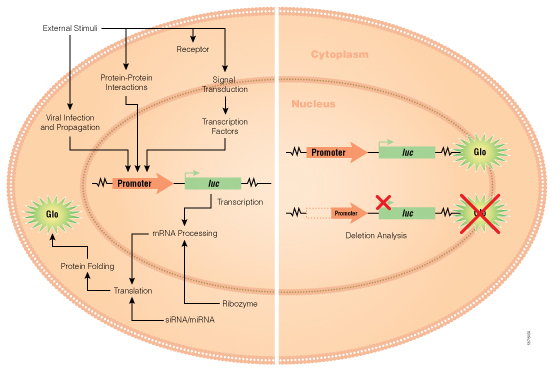
Bioluminescent reporter assays are an excellent choice for analyzing gene regulation because they provide higher sensitivity, wider dynamic range and better signal-to-background ratios compared to colorimetric or fluorescent assays. In a typical genetic reporter assay, cells are transfected with a vector that contains the sequence of interest cloned upstream of a reporter gene, and the reporter activity is used to determine how the target sequence influences gene expression under experimental conditions. A second control reporter encoded on the same or a different plasmid is an essential internal control. The secondary reporter is used to normalize the data and compensate for variability caused by differences in cell number, lysis efficiency, cell viability, transfection efficiency, temperature, and measurement time.
For genetic reporter assays, using a secondary control vector with a weak promoter like PGK or TK to ensures that the control does not interfere with activation of your primary reporter vector. Transfection of high amounts of the control plasmid or putting the control reporter under control of a strong promoter like CMV or SV40 often leads to transcriptional squelching or other interference with the experimental promoter (i.e., trans effects). Reporter assays can also be used to quantitatively evaluate microRNA activity by inserting miRNA target sites downstream or 3´ of the reporter gene. For example, the pmirGLO Dual-Luciferase miRNA Target Expression Vector is based on dual-luciferase technology, with firefly luciferase as the primary reporter to monitor mRNA regulation and Renilla luciferase as a control reporter for normalization.
Here in Technical Services we often talk with researchers who are just starting their project and looking for advice on designing their genetic reporter vector. They have questions like:
- How much of the upstream promoter region should be included in the vector?
- How many copies of a response element will be needed to provide a good response?
- Does the location of the element or surrounding sequence alter gene regulation?