Inflammasomes: A Few Basics
Inflammasomes are protein complexes composed of immune system receptors and sensor molecules. These complexes can respond to both infectious organisms and molecules derived from host proteins. When activated, a series of receptors and molecules signal via either pathogen-associate molecular patterns (PAMPs) induced by microbial pathogens, or danger-associated molecular patterns (DAMPs) induced as a result of endogenous stressors; the common next step in signaling is through pattern recognition receptors (PRR).
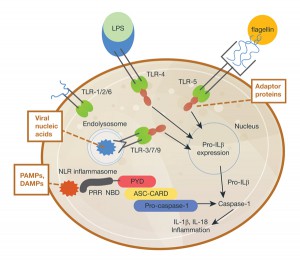
Inflammasome activation is integral to the host immune response in mice and humans (1). The activation results in signaling that activates the caspase-1 scaffold, causing release of immune mediators such as interleukins IL-1β and IL-18. So, whether inflammation is host tissue- or pathogen-induced, inflammasome activation results in a cascade of receptor signaling and mediator release, of which caspase-1 is a critical component.
Inflammasomes in Disease
In their 2015 review, H. Guo et al. (1) write extensively on diseases associated with inflammasomes. A number of metabolic and neurologic disorders that were not originally associated with inflammation are now increasingly recognized as having an inflammatory component. Multiple sclerosis, Alzheimer’s disease and Parkinson’s disease have different disease courses and characterization/symptoms, but share a neurological debilitation caused by chronic tissue attack by rogue immune cells.
Additionally, metabolic diseases including atherosclerosis, type 2 diabetes and obesity, have been connected to inflammasome activation using research models.
Today these metabolic and neurologic diseases comprise some of the largest global public health problems, which translates not only to astronomic financial costs to employers, governments and ultimately you and me, but more importantly, these diseases contribute a substantial threat to longevity and health, resulting in debilitation and even death.
Research Progress
Research on inflammasomes and their activation has teased out much information. Guo et al. present inflammasome signaling diagrams (credited to Debbie Maizels/Nature Publishing Group) that nicely depict what is known about inflammasome activation and signaling.
What are the next steps? Assays and reagents are needed to monitor activation and/or signaling in order to develop and test compounds that block activation or interrupt mediator release, the ultimate goal being to develop therapeutic reagents.
Caspase-1 is an essential component of inflammasome signaling, resulting in the processing and release of cytokines IL-1β and IL-18, and pyroptosis (an immunogenic form of cell death). Detection and monitoring of caspase-1 is thus an interesting target for study of inflammasome activation and ways in which to interrupt signaling.
Until recently, Western blots and ELISA were the primary means by which to monitoring caspase-1 activity. To simplify and provide a more direct means of detecting cell-based caspase-1 activity, Promega scientists developed a sensitive assay that eliminates the need for significant sample processing that is required with these older assays.
How the Caspase-1 Assay Works
The assay employs a one-step format combining a caspase-1 substrate, Z-WEHD-aminoluciferin, with a thermostable luciferase in an optimized, lytic reagent. Assay specificity for caspase-1 can be confirmed by the subsequent use of a caspase-1 inhibitor, Ac-YVAD-CHO.
The assay can be used to measure caspase-1 activity directly in cell cultures or to monitor released caspase-1 activity in culture medium from treated cells. With this assay, caspase-1 activation has been demonstrated in numerous cell culture models of inflammasome activation. Monitoring released caspase-1 activity from culture medium is nondestructive, thereby enabling multiplexing of the biological sample with other assays.
The Caspase-Glo® 1 Inflammasome Assay allows a more efficient and effective assessment of inflammasome activation, as well as enabling high-throughput screening for inflammasome modulators.
We’d love to hear about your caspase-1/inflammasome research. Let us know what you’re working on.
Reference
- Guo, H. et al. (2015) Inflammasomes: mechanism of action, role in disease, and therapeutics. Nature Med. 21 (7), 677–87.
Kari Kenefick
Latest posts by Kari Kenefick (see all)
- What Drives Muscle Fiber Shifts in Obesity and Type ll Diabetes? - October 9, 2025
- How Thalidomide and Molecular Glues Are Redefining Drug Discovery - August 21, 2025
- ATP-Powered Proteins Beyond Kinases – and Why Helicases Are Stealing the Spotlight - August 7, 2025
