This guest blog post is written by Tian Yang, Associate Product Manager at Promega.
In the realm of chemical probe development and drug discovery, understanding the interactions between drugs/compounds and their targets is crucial. Two frequently used metrics to characterize these interactions are IC50 and Kd, which guide researchers in evaluating the potential of compounds in effecting changes in target function. IC50 offers insights into a compound’s potency by quantifying its ability to inhibit a specific biological activity. Kd provides a measure of the affinity between a ligand and its receptor, reflecting how tightly a compound binds to its target (1). Together, these parameters are instrumental in the early stages of drug development, helping to identify promising candidates by assessing a compounds’s binding characteristics and its observed efficacy.
What are IC50 and Kd?
IC50 is a metric used to measure the potency of a substance in inhibiting a specific biological function. It quantifies the concentration necessary to reduce a process, such as enzyme activity, by half its maximum value, offering a direct measure of a compounds’ efficacy under specific experimental conditions. As IC50 is an operational measure derived from fitting experimental data to logistic curves, IC50 can be determined from a wide variety of assays aimed to assess the effect of a drug-like molecule on the target of interest (2).
In contrast, Kd, or the dissociation constant, is a thermodynamic parameter that provides a precise measurement of the binding affinity between a molecule and its target. Representing the concentration at which half of the available binding sites are occupied, Kd offers an intrinsic view of the interaction strength, independent of external variables (1). A number of methods, such as surface plasmon resonance (SPR), isothermal titration calorimetry (ITC), and radioligand binding assay, have been developed to directly measure compound-target interactions and determine the compound’s Kd (3).
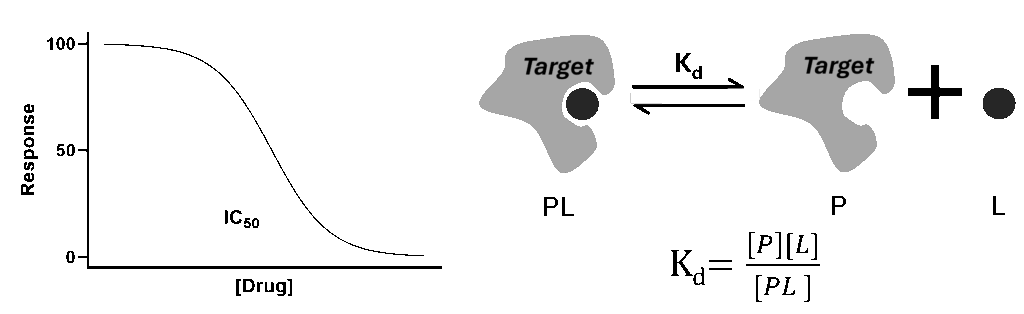
Comparison of IC50 (left) vs. Kd (right)
How are IC50 and Kd different?
Although both IC50 and Kd inform about the compound-target interactions, the two parameters are not directly comparable. Since IC50 is an empirical metric that characterizes the dose-dependent response induced by compound treatment in a specific assay, IC50 reflects the composite effect of the various interactions present in the assay system, including the compound-target interaction, and is influenced by experimental conditions like target concentration and substrate levels. Thus, IC50 measures the functional potency of the compound/drug under the specific experimental setup. However, this sensitivity to experimental conditions complicates the interpretation and comparison of a compound’s potency across studies, as variations in assay setup can lead to significant fluctuations in IC50 values (2).
Since Kd is a direct measurement of the binding affinity between a compound and its target, Kd values are intrinsic to the compound-target interactions. Unlike IC50 measurements, Kd values are unaffected by variations in experimental conditions. This facilitates more straightforward comparisons of compound-target interactions across different studies and across different targets, with the latter enabling compound selectivity determination. As AI and computational models advance, the more accurate and consistent representation of binding affinity provided by Kd can significantly enhance the prediction and optimization of drug candidates, which is crucial for rational drug development (2, 4).
How to relate IC50 to Kd?
While Kd offers a more consistent measure of target binding affinity, directly quantifying compound-target interactions can be challenging. Techniques like SPR and ITC require specialized instruments, while a radioligand binding assay depends on high quality radioligands and uses radioactivity as the readout (3). In probe-displacement assays using either radioligands or fluorescent ligands as probes, the assay outcomes depend on the affinity and concentration of both the probes and compounds of interest and therefore do not directly report the Kd of the compounds (1, 3, 5).
There are relatively simply methods to convert an IC50 determined from a target-compound binding assay to a more consistent Kd value. It is straightforward to determine an IC50 from a target-specific binding experiment, as it is an empirical metric derived from curve fitting of dose-dependent response observed in the binding assay. Due to this ease of measurement, a number of mathematical approaches have been developed to convert IC50 to Kd (1). Methods based on linear regression analysis, such as the Cheng-Prusoff equation and Schild plots, provide relatively simple formulas for determining Kd from the target-binding IC50 values. These methods require several assumptions about the binding assay to hold true. When the number of assumptions about the assay system needs to be reduced, more advanced mathematical solutions for relating IC50 to Kd are available. For biochemical assays where the assay parameters can be tightly controlled and the mechanism of action for drug-target binding is understood, these advanced methods allow for accurate calculation of the Kd of drug-target interactions.
To characterize drug-target interactions inside living cells or organisms, the factors contributing to the compound’s affinity for its target are numerous and can be challenging to assess independently, complicating the correlation of IC50 to Kd. Nevertheless, methods have been developed to determine the apparent affinity (or Kd-apparent) of a drug molecule for its target in live cells. The NanoBRET® Target Engagement assays, which use probe-displacement to measure target-binding in live cells, can be performed to meet the assumptions required for the Cheng-Prusoff equation, allowing for the determination of Kd-apparent from IC50 measurements via a linearized Cheng-Prusoff analysis (6). A theoretical model has also been established for relating the experimental potency value to a compound’s binding affinity in a cellular thermal shift assay, an alternative method for assessing target-binding in live cells (7).
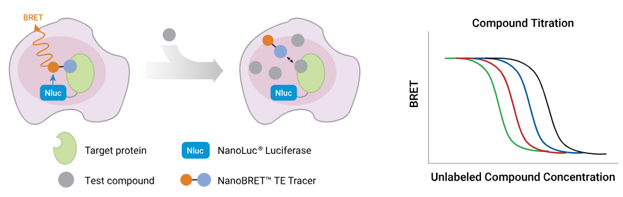
Principle of the NanoBRET® Target Engagement Assay
Summary
While both IC50 and Kd are used to characterize the interactions of compounds/drugs with their biological targets and are pivotal in drug discovery and development, their differences highlight the importance of selecting the appropriate parameter based on the specific goals and contexts of the study. IC50 provides functional potency of a compound under specific experimental conditions and is often easy to measure. However, the assay-specific nature of IC50 makes comparison of IC50 across studies challenging. Conversely, Kd describes the intrinsic binding affinity between a compound and its target that is independent of the experimental setup, facilitating the optimization of compound target binding and compound selectivity. While direct measurement of Kd can be technically demanding, a number of methods have been developed to estimate Kd from IC50 measurements, including for cellular target binding assays. Incorporating both IC50 and Kd into drug discovery can guide development by enhancing the desired compound properties and the understanding of a drug’s mechanism of action, thereby accelerating new drug development.
References:
- Barbet, J. and Huclier-Markai, S. (2019) Equilibrium, affinity, dissociation constants, IC50: Facts and fantasies. Pharm. Stat. 18, 513-525.
- Kalliokoski, T., et al. (2013) Comparability of Mixed IC50 Data – A Statistical Analysis. PLoS One 8(4), e61007.
- Ma, W., et al. (2018) Overview of the detection methods for equilibrium dissociation constant KD of drug-receptor interaction. J. Pharm. Anal. 8(3), 147-152.
- Kramer, C., et al. (2012) The Experimental Uncertainty of Heterogeneous Public Ki Data. J. Med. Chem. 55, 5165-5173.
- Martin, R.L., et al. (1991) A simple method for calculating the dissociation constant of a receptor (or enzyme) · unlabeled ligand complex from radioligand displacement measurements. Arch. Biochem. Biophys. 284(1), 26-29.
- Vasta, J. D.,et al. (2018) Quantitative, wide-spectrum kinase profiling in live cells for assessing the effect of cellular ATP on target engagement. Cell. Chem. Biol. 25, 206–214.
- Seashore-Ludlow, B., et al. (2018) Quantitative Interpretation of Intracellular Drug Binding and Kinetics Using the Cellular Thermal Shift Assay. Biochemistry 57, 6715-6725.
