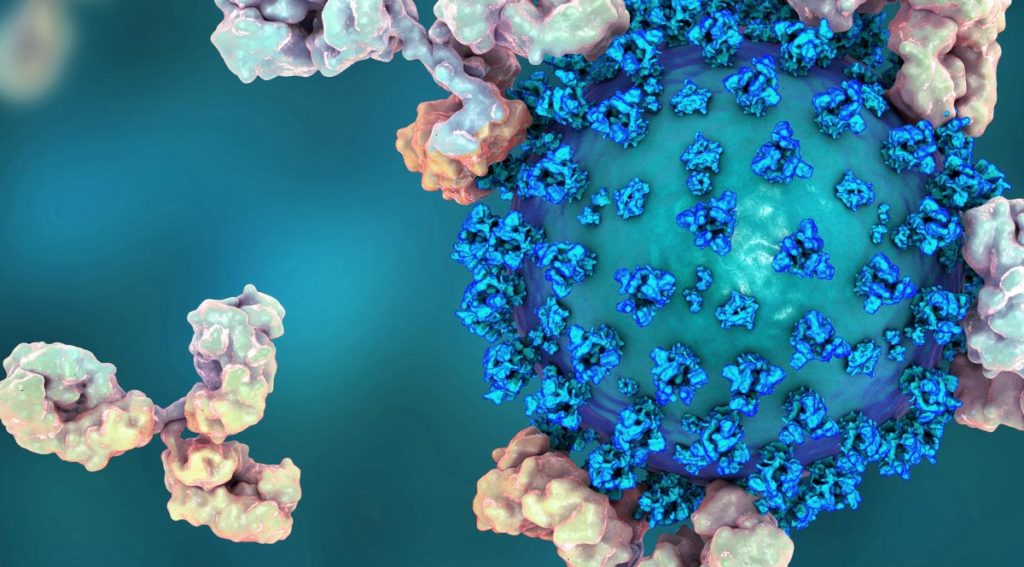
As the SARS-CoV-2 coronavirus continues to spread throughout the world, the race is on to produce antivirals and vaccines to treat and prevent COVID-19. One potential treatment is the use of human monoclonal antibodies, which are antibodies engineered to target and block specific antigens. A recent study by Wang, C. and colleagues published in Nature Communications showed that human monoclonal antibodies can be used to block SARS-CoV-2 from infecting cells.
The authors began with a collection of known antibodies against a virus similar to SARS-CoV-2—its predecessor SARS-CoV (the virus that caused the SARS pandemic in 2002). SARS-CoV-2 exhibits 77.5% sequence identity with SARS-CoV, making it highly likely that an antibody that could bind and block SARS-CoV could have a similar effect on SARS-CoV-2.
The collection of antibodies against SARS-CoV were produced from “humanized” H2L2 mice. Traditionally, antibodies are produced by injecting the antigen into animals, such as rabbits, mice or horses. However, these antibodies contain species-specific regions foreign to humans that could trigger unwanted immune responses, so the antibodies must undergo tedious humanization steps. Humanized mice, on the other hand, have been engineered to produce antibodies with human (instead of mouse) sequences already built in, thus speeding up the antibody production process.
From an existing library of 51 antibodies against SARS-CoV produced from H2L2 mice, the authors found one (named 47D11) that was able to block the infection of SARS-CoV-2. Further characterization showed that 47D11 binds to a conserved receptor-binding region on the surface “spike” proteins of both SARS-CoV and SARS-CoV-2. The spike proteins are what binds to the human cell receptor ACE2, which allows the virus to enter and infect cells.
The authors suspected that 47D11 stopped infection by blocking the binding of the spike proteins to the ACE2 receptor. Surprisingly, this was not the case. Even when 47D11 is bound to the viral spike proteins, interaction with the ACE2 receptor could still occur. The authors therefore concluded that 47D11 works through an unknown mechanism. This result, although unexpected, is good news because it suggests that 47D11 might be used in combination with other antibodies that do block receptor interaction. There is hope that a cocktail of several antibodies that target slightly different aspects of the viral infection pathway can result in an even more effective treatment.
A caveat of this study is that the neutralization effect of the antibody was only tested in cells and not yet in animals or humans. Therefore, more studies are needed to determine whether 47D11 will be effective in human patients. Nonetheless, this study demonstrates the possibility of using human monoclonal antibodies to combat the spread of SARS-CoV-2 and potentially save lives.
Visit our website for resources to support:
SARS-CoV-2 Viral Research
SARS-CoV-2 Serology Testing
Related Posts
Latest posts by Johanna Lee (see all)
- A Live-Cell NanoBRET Assay Shines Light on Toxic RNA–Protein Interactions in Myotonic Dystrophy - February 26, 2026
- Your Science in Review: Our Top Blogs of 2025 - December 30, 2025
- Polyserine Targeting: A New Strategy Against Neurodegeneration - September 30, 2025
