Targeted protein degradation (TPD) is a strategy used to selectively remove proteins from cells, rather than simply blocking their activity. Traditional small-molecule drugs work by binding to a protein and inhibiting its function, leaving the protein intact. In contrast, TPD harnesses the cell waste-disposal system—in particular, the ubiquitin-proteasome pathway—to tag the target protein for destruction. Once tagged, the protein is chopped up and recycled by the proteasome, eliminating it from the cell.
Perhaps the best known TPD approach uses PROTACs (proteolysis-targeting chimeras), which are bifunctional molecules: one end binds the protein of interest, and the other recruits an E3 ubiquitin ligase. By bringing the protein and ligase together, the PROTAC triggers ubiquitin tagging and subsequent degradation.
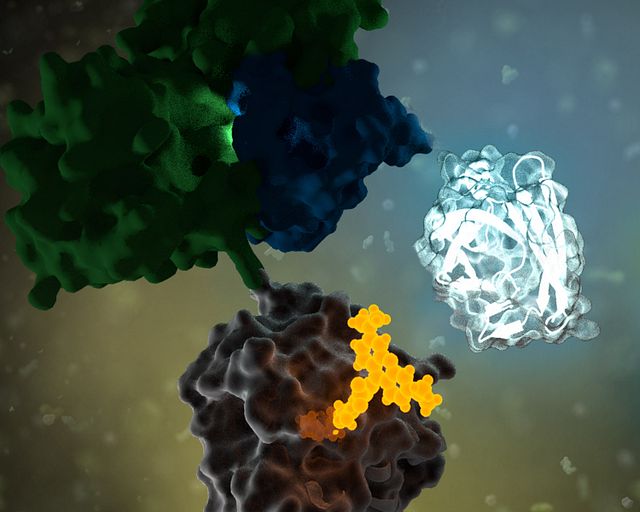
Molecular glues achieve the same end result—selective protein destruction—in a different way. Instead of acting as a physical bridge between the protein and the E3 ligase, molecular glues bind to one protein (often the ligase) and subtly change its shape or surface properties, improving interaction with the target protein. This induced fit causes the target protein to be ubiquitinated without a large, two-part molecule like a PROTAC.
Continue reading “How Thalidomide and Molecular Glues Are Redefining Drug Discovery”

 The ability to manipulate genes and proteins and observe the effects of specific changes is a foundational aspect of molecular biology. From the first site-directed mutagenesis systems to the development of knockout mice and RNA interference, technologies for making targeted changes to specific proteins to eliminate their expression or alter their function have made tremendous contributions to scientific discovery.
The ability to manipulate genes and proteins and observe the effects of specific changes is a foundational aspect of molecular biology. From the first site-directed mutagenesis systems to the development of knockout mice and RNA interference, technologies for making targeted changes to specific proteins to eliminate their expression or alter their function have made tremendous contributions to scientific discovery. 