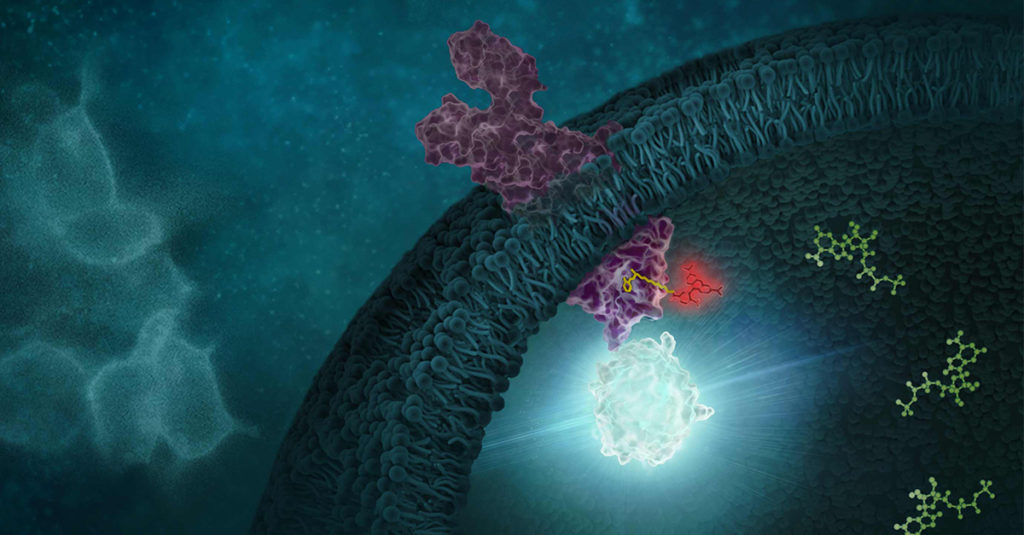
Monitoring and quantifying drug-target binding in a live-cell setting is important to bridging the gap between in vitro assay results and the phenotypic outcome, and therefore represents a crucial step in target validation and drug development (1). The NanoBRET™ Target Engagement (TE) assay is a biophysical technique that enables quantitative assessment of small molecule-target protein binding in live cells. This live-cell target engagement assay uses the bioluminescence resonance energy transfer (BRET) from a NanoLuc® luciferase-tagged target protein and a cell-permeable fluorescent tracer that reversibly binds the target protein of interest. In the presence of unlabeled test compound that engages the target protein, the tracer is displaced, and a loss of BRET signal is observed. Due to the tight distance constraints for BRET, the signal measured is specific to the target fused to NanoLuc® luciferase.
Promega offers over 400 ready-to-use assays for multiple target classes, including kinases, E3 ligases, RAS, and many others. For targets that do not have an existing NanoBRET™ TE assay, Promega offers NanoBRET™ dyes, NanoLuc® cloning vectors, and NanoBRET™ detection reagents to develop novel NanoBRET™ TE assays.
To learn more about the NanoBRET™ TE platform, see the NanoBRET™ Target Engagement Technology Page on our website.
One critical component in the development of novel NanoBRET™ TE assay is the creation of the cell-permeable fluorescent tracers (NanoBRET™ tracers) against the target protein of interest. The tracers are bifunctional, consisting of a NanoBRET™-compatible fluorophore and a target-binding moiety connected by a linker. While the NanoBRET™ 590 dyes have demonstrated consistently robust cell permeability and optimal spectral overlap with NanoLuc® for BRET, a ligand capable of binding to the target protein of interest needs to be identified to generate a NanoBRET™ tracer.
What Are DNA-Encoded Libraries?
DNA-Encoded Libraries, (DELs), have emerged as powerful tools for discovering small molecule ligands to target proteins of interest at an unprecedented scale. . owing to the ability of a DEL to enable the synthesis of larger libraries of compounds and to target proteins without any prior structural knowledge of the proteins or their ligands (2). Because each member of a DEL contains a DNA barcode and a small molecule separated by a linker, DEL is primed for discovering leads within therapeutic modalities that rely on bifunctional chemistry, such as proteolysis targeting chimeras (PROTACs). Since NanoBRET™ tracers are also bifunctional, ligands identified from DEL selections could serve as ideal candidates for developing novel NanoBRET™ tracers that can enable NanoBRET™ TE assays for new targets.
Proof of Concept Hit-to-Assay DEL Workflow
In a recent publication in Cell Chemical Biology, a team of scientists at Promega and WuXi AppTec presented a workflow for converting hits from a DEL screen to BRET probes using aurora kinase A (AURKA) as the target protein (3). From a collection of 16.8 billion compounds pooled from 41 DELs, two representative hits identified in the DEL screen were selected for BRET probe development against AURKA. A panel of BRET probes were synthesized by coupling the functionalized DEL hits to NanoBRET™ dyes with varying linker architectures, and through a systematic evaluation, a cell-permeable BRET probe was identified, and an optimized NanoBRET™ TE assay for AURKA was established to enable quantitative analysis of target binding at AURKA in live cells. In addition, using bromodomain-containing protein 4 and I-doleamine 2,3-dioxygenase 1 as examples, BRET probes were successfully generated from DEL ligands previously reported in the literature, demonstrating that existing DEL databases could be a resource for creating new BRET probes.
Prioritizing DEL Screening Hits with a Live-Cell Target Engagement Assay
Similar to other biochemical screens using purified proteins, predicting the cellular activity of the hits identified from a DEL selection can be challenging. Therefore, a workflow that enables the prioritization of preliminary hits from DEL screens based on target engagement in live cells could benefit the DEL-based drug discovery process. To that end, the team demonstrated that the AURKA NanoBRET™ TE assay can be used to evaluate the binding of the functionalized DEL ligands in live cells, and their results showed that, while functionalized ligands had comparable intrinsic affinities for AURKA, the apparent affinities of these ligands are drastically different in live cells, highlighting the importance of cellular target engagement assays for prioritizing novel hits identified in DEL screens.
Evaluating Panels of Inhibitors with a NanoBRET™ TE Assay
Besides the functionalized DEL ligands, the AURKA NanoBRET™ TE assay with the DEL-derived BRET probe was successfully used to assess a panel of aurora kinase inhibitors representing different chemotypes. Furthermore, the team showed that the DEL-derived BRET probe displayed promiscuity against human kinases in cells, where 40 of the 192 kinases assessed were engaged by the BRET probe, including several understudied kinases. While target specificity is often a premium to chemical probe development and drug discovery, promiscuous BRET probes are beneficial by enabling NanoBRET™ TE assays against multiple target proteins, as the specificity of the NanoBRET™ TE assay is provided by the NanoLuc® tag and the stringent distance constraints for BRET. Consequently, DEL screens that yield chemical probes displaying broad promiscuity may provide the ideal engine for developing novel NanoBRET™ TE assays, and these assays can in turn serve as a platform to validate drug-target engagement in live cells and support lead optimization.
NanoBRET™ Tracer Development from Known Ligands
While the bifunctional nature of the DEL ligands is conducive to design of NanoBRET™ tracers, small molecule ligands or inhibitors against target proteins identified by other technologies have been successfully converted to NanoBRET™ tracers as well. A couple of recent examples are highlighted below.
WD-repeat-containing protein 5 (WDR5) is an important member of the protein complex that regulates histone methylation, an epigenetic modification crucial to transcriptional regulation. Dölle and colleagues (4) sought to develop a PROTAC degrader of WDR5 using two established WDR5 antagonists as anchoring scaffolds. To facilitate the evaluation of cellular permeability of the candidate PROTACs, they developed a new NanoBRET™ TE assay against WDR5, using one of the WDR5 inhibitors and its co-crystal structure with WDR5 to guide the design of the NanoBRET™ tracer. The NanoBRET™ live-cell target engagement assay was then performed in permeabilized, living cells to assess permeability and cellular affinity of the series of PROTACs synthesized targeting WDR5.
Bashore and colleagues aimed to develop chemical probes against Tau tubulin kinase 1 and 2 (TTBK1/2), which have been linked to neurodegenerative diseases and are considered “dark” (understudied) kinases (5). The existing cell-based assays currently used to measure activity of TTBK inhibitors rely on indirect measurement. This study by Bashore et al. describes a new NanoBRET™ tracer against TTBK1/2 that enables direct measurement of cellular target engagement by their compounds. For the target-binding moiety in the new tracer, a small molecule analog prepared for the initial Structure-Activity Relationship (SAR) assessment was used. The resulting NanoBRET™ TE assay enabled by this new tracer was used to profile the cellular affinity of the library of compounds against TTBK1/2. Bashore et al. wrote that:
“Our TTBK1/2 NanoBRET assays offer direct assessment of binding to TTBK1/2 in live cells. Furthermore, these assays allow evaluation of binding kinetics, compound residence time, and cellular penetrance and can be used to drive iterative medicinal chemistry optimization.”
The lead compound was further characterized for its effect on downstream signaling events and activity in phenotypic assays, which supported the potential use of the lead compound as a tool compound for understanding the biological roles of TTBK1/2.
Read more on our blog about how NanoBRET™ Assays are used in research and development.
Summary
The Hit-to-Assay DEL workflow exploits the bifunctional nature of the DEL ligands to facilitate the generation of novel NanoBRET™ tracer, a critical step in enabling live-cell target engagement studies using NanoBRET™ TE assays. The resulting NanoBRET™ TE assays can then be used to prioritize the DEL ligands based on permeability and cellular affinity, potentially accelerating the DEL screening campaigns.
However, developing new NanoBRET™ tracers is not limited to DEL ligands. Small molecule inhibitors previously reported in literature or identified in SAR studies can also serve as a starting point for designing new tracers, as demonstrated in two recent publications highlighted above. These tracers led to NanoBRET™ TE assays for new protein targets, providing a tool to query compound-target interactions for these proteins in live cells.
Literature Cited
- Literature CitedStefaniak, J. and Huber, K.V.M. (2020) Importance of Quantifying Drug-Target Engagement in Cells. ACS Med. Chem. Lett. 11(4) 403-406.
- Satz A.L. et al. (2022) DNA-encoded chemical libraries. Nat. Rev. Methods Primers. 2, 3.
- Teske, K.A. et al. (2023) DELs enable the development of BRET probes for target engagement studies in cells. Cell Chem. Biol. 30, 1–12.
- Dölle, A. et al. (2021) Design, Synthesis, and Evaluation of WD-Repeat-Containing Protein 5 (WDR5) Degraders. J. Med. Chem. 64(15) 10682-10710.
- Bashore, F.M. et al. (2023) Modulation of tau tubulin kinases (TTBK1 and TTBK2) impacts ciliogenesis. Sci. Rep. 13, 6118.
