Mention the word penicillin and it conjures up images of mold growing on bacterial culture plates and Dr. Alexander Fleming observing that the mold had killed the surrounding bacteria, ushering in the age of antibiotics. Bacterial infections could easily be treated with penicillin or any one of the bewildering array of new antibiotics continually being discovered. The result of using these antimicrobial drugs: numerous lives were saved and human health improved. However, bacteria are clever organisms and as quickly as humans developed an antibiotic to treat infection, the microbes would find a way around the bacteriostatic or bacteriocidal compound. It is a scary world where antibiotics are rendered impotent and fewer and fewer weapons are left in the arsenal to treat multidrug-resistant Staphylococcus aureus (MRSA) and hospital-acquired drug-resistant Gram-negative bacteria (e.g., Acinetobacter baumannii).
One place scientists have turned for the next potential antibiotic is bioinformatics and the analysis of sequenced genomes. A recently completed genome included the anaerobic Gram positive bacterium, Clostridium cellulolyticum. It was originally isolated from decaying grass compost, and its claim to fame is as a model organism known for its cellulolytic ability, meaning the bacterium breaks down cellulose. Interestingly, C. cellulolyticum under standard growth conditions had never produced secondary metabolites. However, mining of its genome found genes for biosynthesis of secondary metabolites. Therefore, the scientists from Leibniz-Institut für Naturstoff-Forschung und Infektionsbiologie in Jena, Germany, attempted to find alternative conditions to induce biosynthetic pathways and see what product would result. They tried different growth supplements, including a variety of carbon sources as well as different stressors (e.g., temperature, heavy metals or starvation) but nothing was detected in the extracts from these treated cultures.
The next idea was to expose the bacterium to a more natural environment. Since C. cellulolyticum was found in compost, the researchers added an aqueous extract from soil to the growth medium prior to inoculation. Analysis by reverse-phase (RP)-HPLC found new peaks that could not be explained by the soil supplement alone. Furthermore, these new peaks could be reproducibly detected using soil samples from different sources (e.g., compost or soil core samples).
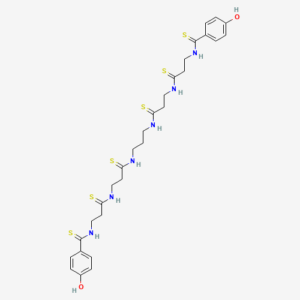
Analysis of the major metabolite, named compound 1, involved isolating a pale yellow compound, characterizing its chemical formula (C29H38N6O2S6) and subjecting it to NMR spectroscopy, yielding a symmetrical molecule. Using high resolution mass spectrometry (HRMS) and spectral analysis, the sulfur atoms present were resolved into a thioamide grouping. The newly discovered compound is a unique symmetrical polythioamide called closthioamide, (Scroll down the abstract to see the structure.) Not only is the symmetry of the polythioamide molecule unusual, but only four of the 170,000 known natural organic compounds feature this functional group, and the authors express amazement at having a demonstrated polythioamidated metabolite.
The newly dubbed closthioamide was tested in a standard antimicrobial assay and found to be active against MRSA and vancomycin-resistant enterococci (VRE). Further testing revealed that the thioamide moieties were the key to the molecule’s antimicrobial functionality. A similar molecule was synthesized where the sulfur was replaced with oxygen and tested in the same assay; it had no antibiotic activity against the same panel of bacteria.
While finding and triggering silent biosynthetic pathways to generate new and interesting natural products is an old technique, this paper probed the largely unexplored secondary metabolites of an anaerobic bacterium. In this case, C. cellulolyticum produced an unusual symmetrical molecule with antibiotic activity. Not bad for a microorganism better known for its catabolism of cellulose than its ability to synthesize interesting metabolites. The strategy undertaken in this paper highlights both the utility of bioinformatics analysis of sequenced genomes and exploring what secondary metabolites of anaerobic bacteria might offer us in our continuing battle against undesirable bacterial colonization of humans and other mammals.
Lincke T, Behnken S, Ishida K, Roth M, & Hertweck C (2010). Closthioamide: An Unprecedented Polythioamide Antibiotic from the Strictly Anaerobic Bacterium Clostridium cellulolyticum. Angewandte Chemie (International ed. in English), 49 (11), 2011-2013 PMID: 20157900
Sara Klink
Latest posts by Sara Klink (see all)
- A One-Two Punch to Knock Out HIV - September 28, 2021
- Toxicity Studies in Organoid Models: Developing an Alternative to Animal Testing - June 10, 2021
- Herd Immunity: What the Flock Are You Talking About? - May 10, 2021

It’s a great find you describe here, but how long did it take the researchers take to find this? And how long will it take bacteria to ‘evolve’, to alter their genetic makeup? Not so long I think, since they replicate pretty fast..
Other than that, how long ago was it that scientists have found a new antibiotic or cure against MRSA and other multi-resistant bacteria? It’s not likely that it will happen again before the bacteria have found a way to still do their work.
Have you ever heard of bacteriophages? They’re tiny little viruses that enter a bacterium (a different phage for each type of bacterium, and they evolve a long with them) and destroy it within.. a short period. It’s worth a look, I’d say. Check out the BBC documentary ‘Phage, the virus that cures’.
Here’s a link:
http://video.google.co.uk/videoplay?docid=8887931967515748990#
hi Sara…it was great reading your abstract. I want to know more about your discovery. How exactly you did all this etc etc…n more about the drug…can it be developed into broad spectrum antibiotics?? I am studying pharmacy right now so i m keen to know about your discovery…
please contact me as soon as possible…mail me at 2009dixitba@gmail.com