Part one of three
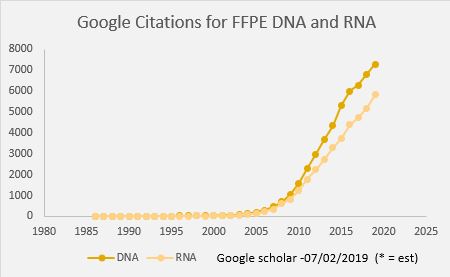
Formalin Fixed Paraffin Embedded samples (FFPE) have been a mainstay of the pathology lab for over 100 years. Initially FFPE blocks were sectioned, stained with simple dyes and used for studying morphology, but now a variety of biomolecules can be analyzed in these samples. Over the past 10 years we have discovered that there is a treasure trove of genomics data waiting to be unearthed in FFPE tissue. While viral RNAs and miRNA were some of the first molecules found to be present and accessible for analysis starting in the 1990s, improvements to DNA and RNA extraction methods have demonstrated that PCR, qPCR, SNP genotyping, Exome and WGS are possible. This has resulted scientific publications of DNA and RNA data generated from FFPE samples starting in 2006, and today we see immense amounts of data generated from FFPE—with nearly 2000 citations in 2018 reporting sequencing of FFPE samples.
Depending on the type of project, prospective or retrospective, the genomics scientist has an opportunity to affect the probability of success by better understanding the fixation process. The challenge with FFPE is the host of variables that have the potential to negatively affect downstream assays.
The biobanking field through its association, The International Society of Biobanks and Environmental Repositories (ISBER), offers recommendations for collection and storage of FFPE samples and offers recommendations and proficiency program on its website: www.isber.org. In addition, biobanking scientists have identified preanalytical factors that can affect the utility of samples for downstream assays. In a recent review article, Bass et al. (2014) provide some guidance for scientists wishing to perform molecular analysis of FFPE samples. The authors review exiting literature and examine pre-analytical variables and their affect on downstream analysis. Table 1 from the article outlines the factors that were reviewed. For more details on their findings, we recommend reading the entire article.
When the samples are processed promptly and with a defined workflow, results can be quite good from FFPE samples.
While there are many individual preanalytical factors, there are two broad categories that stand out: handling of samples prior to the fixation process and the fixation protocols and procedures. Some of these can be controlled in the lab, while others may be outside of the lab’s direct control. In speaking with scientists using FFPE in prospective studies, labs that have the most success work closely with a partner in the pathology department to control sample processing and the fixation process. Specifically, they work to procure samples rapidly and timing of fixation. When the samples are processed promptly and with a defined workflow, results can be quite good from FFPE samples.
Retroactive studies or studies where samples are procured from many locations can be more challenging since there is less control and likely more variability between individual specimens. For these samples choices on how to determine quality and QC DNA or RNA for suitability in assays becomes much more important. In my next blog, we’ll review methods to quantitate or QC FFPE samples and the information that different tests can provide to help you decide if a sample is worth using.
