At the time that I’m writing this, I still haven’t succumbed to the “yuck” that’s been knocking out my co-workers one-by-one since November. Those of us who are still healthy were discussing how we fortify our immune systems in preparation for the flu season. All of the suggestions were pretty typical—orange juice, Vitamin C supplements, and of course, the the annual flu shot.
For all of the agencies responsible for the production of the seasonal influenza vaccine, preparation for flu season begins long before the rest of us are stocking up on Emergen-C. Early in the year, virus surveillance programs begin compiling data to predict the following year’s flu season. In the United States, this is orchestrated by the Epidemiology and Prevention Branch in the Influenza Division at Centers for Disease Control and Prevention (CDC). They continuously process data submitted from all over the country by both public health and clinical labs. Clinical lab data helps CDC understand the timing and intensity of the flu, while the public health labs provide information on which viruses are circulating and which populations are affected. This data helps predict which strains will be most common in the upcoming flu season. CDC also receives statistics from health care providers and hospitals about hospitalization and mortality rates, which helps predict which populations are most vulnerable, and to what extent.
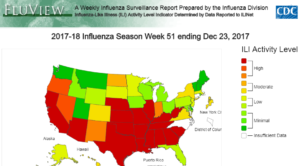
These studies also help inform the development of the annual influenza vaccine. The vaccine usually contains two strains of Influenza A and one of Influenza B. The specific strains are selected by the World Health Organization in a consultation with the directors of the WHO Collaborating Centers for Reference and Research on Influenza. This panel includes CDC, The Francis Crick Institute in London, Victoria Infectious Diseases Reference Laboratory in Melbourne, National Institute for Infectious Diseases in Tokyo and National Institute for Viral Disease Control and Prevention in Beijing. Together, they review all of the surveillance data and clinical studies and form a recommendation for which three strains to include in the vaccine that year. Those recommendations are then reviewed by the regulatory agencies of each country, who make the final decisions about the composition of the vaccine to be manufactured in that country.
In the United States, the final decision rests on more than just epidemiological factors. There must be a vaccine virus available that is similar enough to each of the recommended strains to produce immunity. Assuming such a vaccine exists, it also must be suitable for mass-production under regulatory-approved conditions. Chicken egg-based manufacturing is the dominant method in the United States, though it isn’t always compatible with recommended strains (This is also why you are usually asked if you are allergic to eggs when you get your flu shot.) For example, H3N2 grows poorly in eggs, so H3N2 vaccines must be produced with other methods. In the United States, the first approved cell-based vaccine was manufactured in 2016, and some recombinant flu vaccines are beginning to creep into the market.
Once an official vaccine receives approval, private sector manufacturers begin inoculating eggs with the chosen viruses and harvesting the fluid after incubation. The viruses are either killed for the flu shot or weakened for the nasal spray. After testing and approval, as many as 166 million doses will be released for public vaccination. A few hundred of those will end up at the Promega Wellness Center, where we all line up in hopes of avoiding this wretched “yuck.”
Of course, if the “yuck” is really just a rhinovirus or coronavirus that is making the rounds, probably the best we can do is wash our hands with soap and water, stay home when we are sneezy and wheezy and get plenty of rest and fluids.
Here’s to a healthy 2018!

