Cannabinoids. What are they? Sometimes, Wikipedia can give a nice definition:
Synthetic cannabinoids (SCs) were originally created for the scientific investigation of two cannabinoid receptors, CB1 and CB2, but have made their way to the streets as “safe” and “legal” alternatives to marijuana.
The problem is that these SCs engage the cannabinoid receptors more completely and with higher affinity than anything derived from marijuana. As a result, SCs can produce serious side effects that often require medical attention. In fact, you are 30 times more likely to seek emergency medical attention following the use of an SC than with natural cannabinoid sources like marijuana.
SCs are the largest group of compounds currently monitored in Europe by the EU Early Warning System for new psychoactive substances. Regulatory groups around the globe are concerned with the rise of SCs and are beginning to shift definitions for illegal substances away from chemical structure-based descriptions to more biological activity-based definitions.
Assays are needed to provide assessment of bioactivity and, in a recently accepted publication, researchers from the Laboratory of Toxicology (Ghent University, Belgium) and the Laboratory of Forensic Toxicology (University of Freiburg, Germany) collaborated on the development of a bioassay to profile the activity of synthetic cannabinoids and their metabolites on the cannabinoid receptors (1).
CB1 and CB2 are GPCRs coupled to the G0/i protein, leaving many options available to create the bioassay, such as focusing on the internalization of the receptor from the cell surface, GTP binding by the G0/I protein or changes in cAMP and Ca2+. Each method evaluated had drawbacks, being too labor intensive or prone to fluorescence interference by the test compounds or requiring specialized equipment or the use of radioactivity. The authors provide an extensive discussion of the advantages and disadvantages of the various commercially-available systems to monitor GPCR activation.
The final assay was designed using the NanoLuc® Binary Technology (NanoBiT™) (2). The authors like the flexibility, simplicity and sensitivity offered by the system. The NanoBiT™ Assay is a protein complementation assay composed of a large subunit (17.6kDa or LgBiT) and a small subunit (11aa or SmBiT). LgBiT and SmBiT are fused to interacting proteins and, if those two proteins interact, the LgBiT and SmBiT are brought together to reform an active NanoBiT™ Luciferase. The affinity of LgBiT and SmBiT were engineered to be low so that the binding partners are truly needed to bring them together.
The authors tested the LgBiT and SmBiT at the C-terminus of both receptors along with the corresponding SmBiT and LgBiT at the N- and C-terminus of β-arrestin 2 looking for the best combination. β-Arrestin 2 interacts with many GPCR receptors to internalize the receptor and desensitize the cell. The CB1 receptor assay worked best with LgBiT at the C-terminus combined with SmBiT at the N-terminus of β-arrestin 2. The CB2 receptor worked best with SmBiT at the C-terminus paired with LgBiT at the N-terminus of β-arrestin 2.
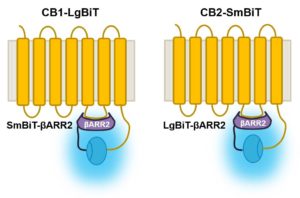
The NanoBiT™ Assay easily monitored agonist activities producing EC50 values in line with the literature values for the natural and synthetic cannabinoids. The assay could also assess the bioactivity of SC metabolites and measure the effects of receptor antagonists. The authors were pleased to report that they could perform the NanoBiT™ Assay with human-derived urine samples and employ the non-lytic assay in real-time from 0-120 minutes as signal begins as soon as they add the test compound. The authors concluded that in the future this assay may be used as a first line screening tool for SCs.
- Cannaert, A. et al. (2016) Detection and activity profiling of synthetic annabinoids and metabolites with a newly developed bio-assay. Analytical Chemistry. Oct 25, Epub ahead of print.
- Dixon, A.S. et al. (2016) NanoLuc complementation reporter optimized for accurate measurement of protein interactions in cells. ACS Chem. Biol. 11, 400-8.
Updated April 9, 2024 to correct broken links.

Dear Kyle,
Thanks for this nice article about my publication where I use the NanoBiT system. A small remark, the optimal combination for the CB2 receptor to measure β-arrestin 2 recruitment is when the CB2 receptor is linked to the SmBiT and the LgBiT is attached in front of the β-arrestin 2. For CB1 is the other way around, as mentioned correctly in your article.
Kindest regards,
Annelies Cannaert
Hello Annelies,
Thank you for the kind words. I apologize for messing up the description of your CB2 construct. I will correct it as soon as possible.
Sincerely,
Kyle