Promega carries a large array of luminescent-based assays to measure cellular events such as viability, cytochrome P450 activity and apoptosis. Recently, we launched a new universal, homogeneous, high-throughput screening method called the ADP-Glo™ Kinase Assay, which measures kinase activity by quantifying the amount of ADP produced during a kinase reaction. While we already offer the Kinase-Glo™ Assays for assessing the quantity of ATP remaining after a kinase reaction, these assays are not ideal for use with low-activity kinases.
The ADP-Glo™ Kinase Assay can monitor the activity of nearly any ADP-generating enzyme with a sensitivity of 0.2pmol/20μl reaction, and can linearly detect ADP generated in a reaction containing up to 1mM ATP. Because the ADP-Glo™ Kinase Assay has a high signal-to-background ratio, it is useful for enzymes with low ATP turnover. Furthermore, the assay features a highly stable luminescent signal, which is ideal for processing multiwell plates in batch mode. The proprietary reagents and luminescent detection means the ADP-Glo™ Kinase Assay is less susceptible to interference from library compounds.
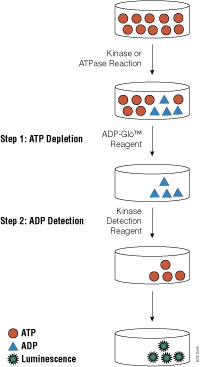
The assay is simple to use: perform your kinase assay, add an equal volume of ADP-Glo™ Reagent to stop the kinase reaction and deplete the remaining ATP; then add twice the initial volume of Kinase Detection Reagent to simultaneously convert ADP to ATP and measure the newly generated ATP in a luciferase reaction. The light output measured directly correlates to the enzyme activity in the kinase reaction.
For more information on the ADP-Glo™ Kinase Assay, visit our Web site or read the Technical Manual.
Sara Klink
Latest posts by Sara Klink (see all)
- A One-Two Punch to Knock Out HIV - September 28, 2021
- Toxicity Studies in Organoid Models: Developing an Alternative to Animal Testing - June 10, 2021
- Herd Immunity: What the Flock Are You Talking About? - May 10, 2021
