Antibody drug conjugates (ADCs) are a new class of therapeutic drugs that uses antibodies to deliver highly toxic drug molecules specifically to the cancer cells. A key requirement for ADCs is the ability of antibody to bind to the cancer cells followed by internalization and subsequent release of drug inside the cells leading to cell apoptosis.
Traditionally, selection of lead antibody candidates for ADCs was done in a sequential workflow where antibodies were first selected based on their affinity followed by characterization involving antibody internalization and drug conjugation. However, there is evidence that high affinity doesn’t always correlate with good internalization and hence there is a need to screen antibodies for internalization properties in addition to their affinities.
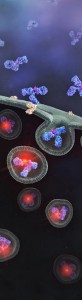
Promega has developed a method that allows antibody to be screened for their internalization properties in a simple, plate-based format. The method uses pH sensor dyes (pHAb dyes), which are not fluorescent at neutral pH but become highly fluorescent at acidic pH. When antibody conjugated with pHAb dye binds to its antigen on the cancer cell membrane they are not fluorescent but upon internalization and trafficking into endosomal and lysosomal vesicles the pH drops and dye becomes fluorescent.
Fluorescence signal, for pHAb dyes conjugated using either amine or thiol chemistry, is minimal at pH>7 and increase significantly as the pH drops to pH 5.0, which is a typical pH in cell endosomal compartment. Moreover, pH response of free pHAb dye is similar to that of conjugated dye indicating that conjugation chemistry doesn’t influence the pH response of the dye.
Due to the high signal-to-background ratios of the dyes, plate-based internalization assays can be performed, enabling screening of large libraries of antibodies for their internalization properties, hopefully leading to improved identification of lead candidates for ADC applications.
