We rely on insulin supplied by our pancreas at the right dose and at the right time to control our blood glucose levels and energy storage. Insulin works by regulating the energy usage of various cell types in the body. When this process goes awry, it can cause diabetes.
There are two types of diabetes, defined by how insulin is dysregulated. In Type 1 Diabetes (T1D), the pancreas produces too little insulin. Patients need to give themselves insulin in order to respond to glucose in the diet. In Type 2 Diabetes (T2D), patients do not respond well to the insulin produced in their body. Therefore, they need to give themselves more to avoid hyperglycemia (high blood glucose).
Medical professionals have been treating diabetic patients with some form of insulin therapy for nearly 100 years. You may know a family member who takes the hormone daily, or have seen a colleague with an insulin pump. Researchers are evaluating different forms and delivery methods of insulin to make it even easier for the many people that rely on insulin. An oral pill could be a pain-free method for patients and make insulin injections a thing of the past.
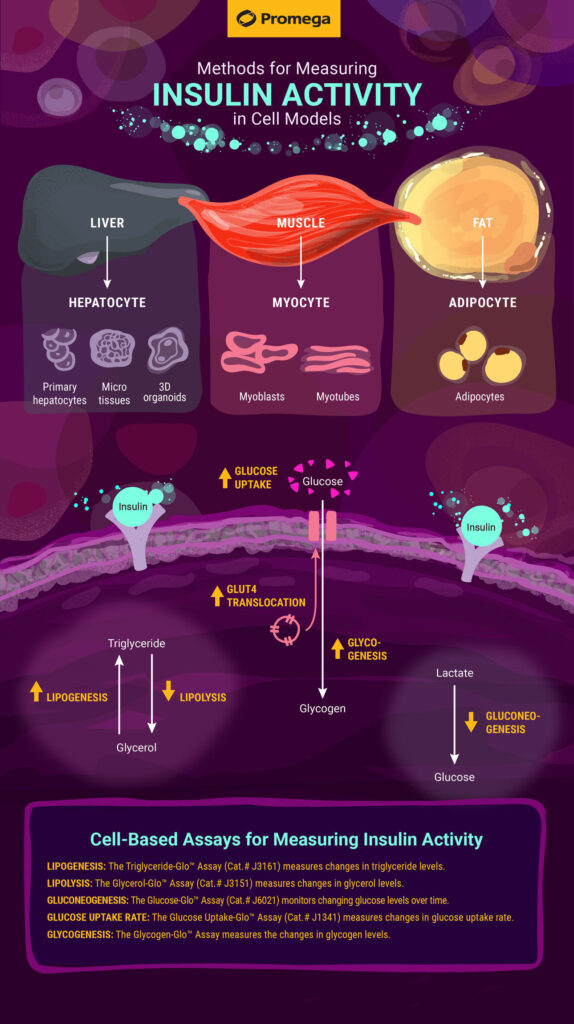
Because of the rise in diabetes and obesity, research related to insulin action continues to grow. Researchers want to better understand the energy usage regulation by insulin. Tools that help researchers include cell-based assays, where specific cell types are grown in the lab and different responses to insulin are measured. In the case of insulin research, three types of cells that are often used are liver cells (hepatocytes), muscle cells (myocytes) and fat cells (adipocytes).
The infographic below illustrates some common pathways that are measured in response to increased insulin levels. Learn about the various metabolic assays from Promega that can help you get a complete picture of insulin activity.
Lipogenesis: The generation of triglycerides and other lipid molecules is upregulated in adipocytes and hepatocytes in response to insulin. In this way, insulin acts as a storage-promoting hormone. Elevated levels of insulin indicate to cells that energy (glucose) is plentiful and should be stored. The increase in triglyceride levels can be measured using the Triglyceride-Glo™ Assay.
Lipolysis: Lipolysis is the breakdown of triglycerides into a molecule of glycerol and three fatty acids. Insulin inhibits lipolysis, so that cells preferentially use the glucose present when insulin levels are high. The result of lipolysis is an increase in glycerol molecules, which can be measured with the Glycerol-Glo™ Assay.
Gluconeogenesis: The generation of new molecules of glucose is inhibited in cells where insulin levels are elevated. Because insulin is elevated in conditions where glucose is elevated, insulin signals cells to not make any more glucose and rather use the glucose present. Levels of glucose can be measured with the Glucose-Glo™ Assay.
Glucose Uptake Rate: Insulin increases the rate at which cells take up glucose through glucose transporters. This allows cells to uptake glucose for storage and use. The Glucose Uptake-Glo™ Assay can be used to measure the increase in rate of glucose uptake.
“Promega products are reliable and can be purchased without any hesitation. My experiment went very well as expected and we have more plans to conduct uptake experiments using the Glucose Uptake-Glo™ Assay.”
Dr. Vivek Pandey, Postdoctoral Scholar, University of Kentucky
Glycogenesis: Glycogen, a polymer of glucose, is a primary carbohydrate storage form. Insulin upregulates the storage of glucose in the form of glycogen in myocytes (muscle cells) and hepatocytes. The Glycogen-Glo™ Assay* can be used to measure the increase in glycogen.
*The Glycogen-Glo™ Assay is currently available through our custom manufacturing service.
See our full portfolio of Metabolic Detection Assays.
This blog was written in collaboration with Maggie Bach, Product Manager.
Latest posts by Johanna Lee (see all)
- Tiny Edits, Big Harvests: How a Massive Mutation Screen Could Transform Crop Engineering - May 21, 2026
- A Live-Cell NanoBRET Assay Shines Light on Toxic RNA–Protein Interactions in Myotonic Dystrophy - February 26, 2026
- Your Science in Review: Our Top Blogs of 2025 - December 30, 2025