The brain is one of the most complex and fascinating parts of biology. Thankfully, it’s also remarkably good at protecting itself. When exposed to a pathogen, an injury or even misfolded proteins, microglia and astrocytes function as the central nervous system’s (CNS) primary immune defenders. They mount an inflammatory response by releasing cytokines and working to contain the damage. Yet this same system can malfunction or not resolve, which manifests as devastating consequences.
Chronic neuroinflammation is now recognized as a shared characteristic across some of the most common and difficult-to-treat neurological conditions. A 2023 review in Signal Transduction and Targeted Therapy highlighted the dualistic nature of neuroinflammation: while acute responses serve a protective role, chronic or dysregulated inflammatory signaling can initiate and accelerate neurodegeneration, identifying these pathways as priority targets for therapeutic intervention (Zhang et al., 2023). A 2025 review in Science reinforced this view, noting that within Multiple Sclerosis, disease-modifying therapies targeting neuroinflammation have seen the most clinical success (Shi & Yong, 2025). This could suggest applications within neurological conditions where the same inflammatory mechanisms are at work.
Understanding how and where these inflammatory signals originate in the CNS is an active area of preclinical research. One cytokine being actively studied is IL-6. IL-6 is produced by several cell types, including astrocytes and microglia in the CNS. As a key mediator of inflammatory responses, it mediates pro-inflammatory effects through its trans-signaling, which occurs via soluble IL-6 receptors. Dysregulation of this mechanism may contribute to the chronic neuroinflammation seen in several neurological conditions. Characterizing how and when IL-6 is secreted from CNS cells is an important step toward understanding the neuroinflammatory processes underlying these disorders.
Limitations with Traditional ELISA
Successful measurement of inflammation has been around since the 1970s, with the invention of the ELISA (Enzyme-Linked Immunosorbent Assay), which enabled detection using antibodies. This approach, although widely used, has some drawbacks. These mainly include multiple wash steps, slow protocols and challenges with sensitivity.
To address these challenges, Promega developed a no-wash, bioluminescence method for detecting target molecules under 70 minutes, the Lumit® Immunoassay. Lumit® Immunoassays are based on NanoLuc® Binary Technology (NanoBiT®). In Lumit® Immunoassays, antibodies or antigens are chemically labeled with the small (SmBiT) and large (LgBiT) subunits of NanoBiT® luciferase. In the presence of the target molecule, the two subunits are brought into proximity, allowing SmBiT and LgBiT to form an active enzyme and generate a bright luminescence signal. This signal can be detected on a microplate reader.
Application of Lumit® Immunoassays for Studying Neuroinflammation
Below we highlight two experiments that utilized the Lumit® Immunoassay platform to detect IL-6 secretion from either microglia or astrocytes.
Experiment 1: Microglia Inflammatory Responses
We thawed microglial cells and grew them on a Poly-D-Lysine coated plate for either 5 or 7 days. We then treated the microglia with 100ng/mL of lipopolysaccharide (LPS) for 1 day and collected conditioned medium.
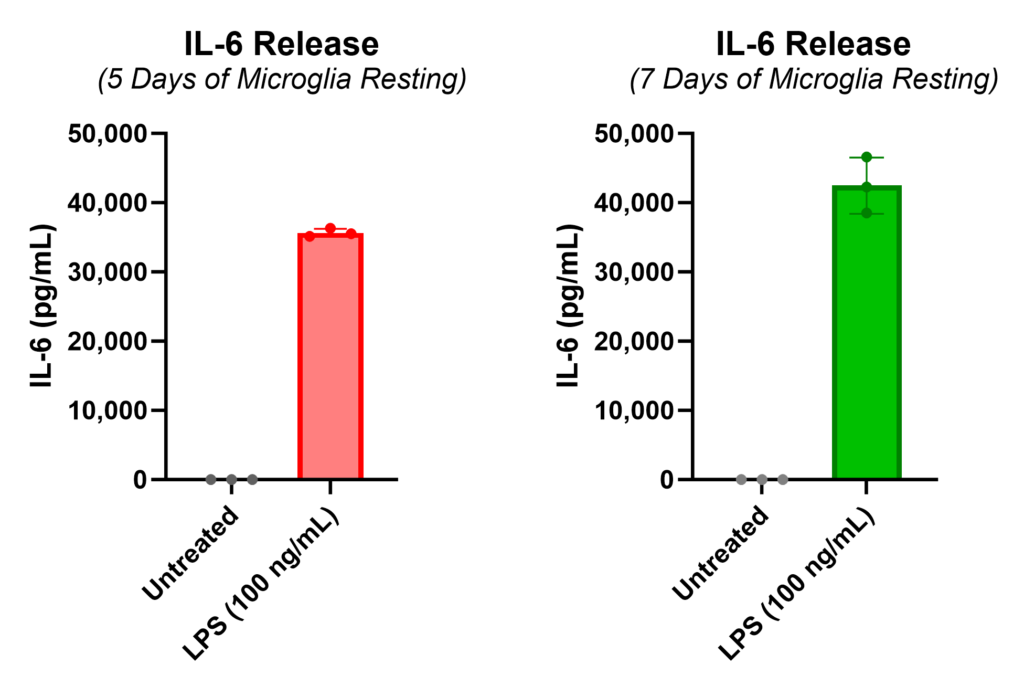
The Lumit® IL-6 immunoassay detected IL-6 in the culture media after LPS treatment. We measured luminescence on the GloMax® Explorer. All IL-6 quantification shown (pg/mL) was based on the IL-6 standard curve generated in the Lumit® assay, and the concentrations of the treated samples were extrapolated from that standard curve. A robust response of released IL-6 was measured after 5 days of resting (red bar) or after 7 days of resting (green bar).
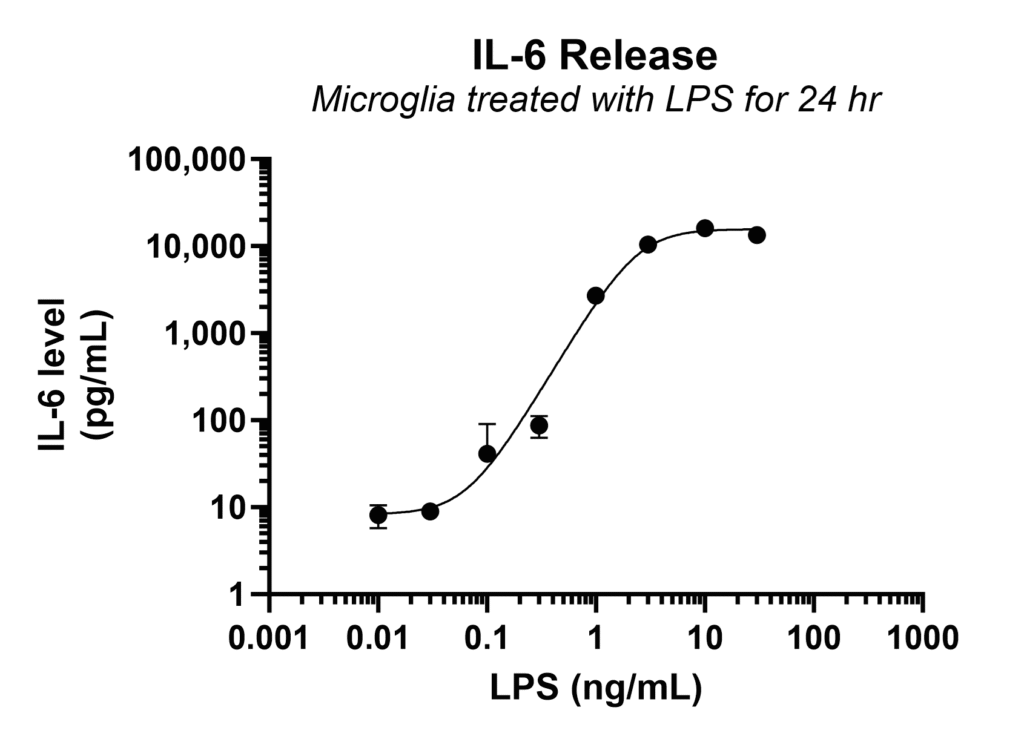
Microglia treated with various doses of LPS showed a clear, concentration-dependent increase in IL-6 secretion, demonstrating the sensitivity of detection in this model.
Experiment 2: Astrocytes Inflammatory Responses
We plated astrocytes derived from iPSCs at 10,000 cells per well and allowed them to rest for several days with periodic media changes before treating with 10ng/ml of tumor necrosis factor alpha (TNF-α). After 48 hours of treatment, we collected the conditioned medium and ran the Lumit® IL-6 immunoassay, measuring luminescence on the GloMax® Explorer.
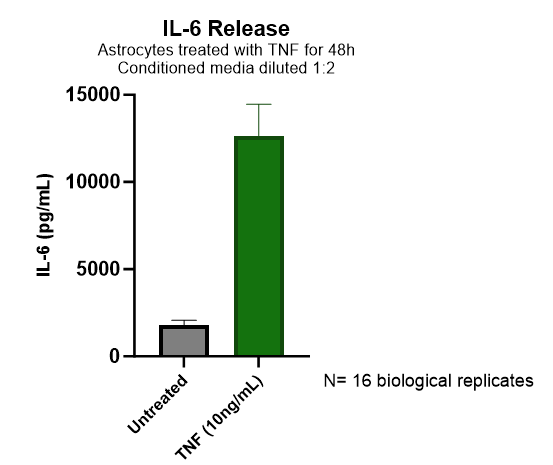
Quantified IL-6 release from astrocytes was greatly increased upon treatment with TNF-α (green bar). All IL-6 quantification shown (pg/mL) was based on the IL-6 standard curve generated in the Lumit® assay, and the concentrations of the treated samples were extrapolated from that standard curve.
Conclusion
If you’re studying neuroinflammation, the Lumit® platform offers a fast, no-wash approach to detecting cytokines at physiologically relevant concentrations. The Lumit® Immunoassay portfolio enables detection in under 70 minutes, without wash steps, making it well suited for the kinds of low-level signaling seen in CNS cell models. With growing evidence that inflammatory signaling plays a role in neurological disorders, the Lumit® platform is a practical fit for early research and drug discovery workflows.
Interested in learning more about the Lumit® platform? Visit our technology page.
Explore the full portfolio of inflammation products.
Citations
Shi, F.D. and Yong, V.W. (2025) Neuroinflammation across neurological diseases. Science 388, 6753.
Zhang, W. et al. (2023) Role of neuroinflammation in neurodegeneration development. Signal Transduction and Targeted Therapy 8, 1486.
Latest posts by Simon Moe (see all)
- Detecting Neuroinflammation in Microglia and Astrocytes - March 19, 2026
- Your Media Choice Might Be Designing Your T-Cell Fate - January 15, 2026
- Insights from 3D Liver Models: Rethinking Fatty Liver Disease with Hormone Correction - October 29, 2025
