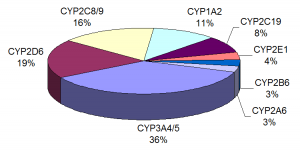
The pregnane X receptor (PXR) is a nuclear receptor known to regulate expression of cytochrome P450 (CYP450) drug-metabolizing enzymes (1). PXR has even been designated the “master xenosensor” due to its ability to upregulate cellular levels of a variety of drug-metabolizing enzymes in response to drugs and foreign chemicals. Elevated levels of CYP450 enzymes can elicit alterations in the pharmacokinetics of co-administered drugs, which can result in adverse drug-drug interactions (DDI) or diminished bioavailability. By assessing PXR activation and CYP450 enzyme induction early in the drug development process, many companies hope to reduce late-stage clinical failures and minimize the high costs associated with bringing a new drug to market.
A paper by Shukla et al. (2) examined over 2,800 clinically used drugs for their ability to activate human PXR (hPXR) and rat PXR (rPXR), induce human cytochrome P450 3A4 enzyme (CYP3A4) at the cellular level, and bind hPXR at the protein level. Several studies have identified PXR as playing a key role in regulating the expression of CYP3A4, an enzyme involved in the metabolism of more than 50% of all drugs prescribed in humans. Since PXR activation and CYP3A4 induction have an impact on drug metabolism and pharmacokinetics, the authors wanted to obtain data that would be valuable in understanding structure-activity relationships (SARs), the connection between chemical structure and biological activity, when prioritizing new molecular entities (NMEs) for further in vitro and in vivo studies.
Continue reading “Screening for Drug-Drug Interactions with PXR and CYP450 3A4 Activation”