
“20 years ago, when I first heard about the creation of human embryonic stem cells, I knew that this was the future. I immediately requested the cells from Dr. Thomson and dropped almost everything else we were doing in our lab. It has been my focus to this day.” The person presenting is Dr. David Russell, a professor at the University of Washington. He is just one of the hundreds of researchers gathered at the BioPharmaceutical Technology Center Institute (a nonprofit supported by Promega) in Madison, Wisconsin for the 13th Annual Wisconsin Stem Cell Symposium that happened this week. This year, it’s not just a symposium, but also a celebration—it’s the 20-year anniversary of the first-ever isolation and culture of human embryonic stem cells (ES cells).
In 1998, Dr. James Thomson, at the University of Wisconsin-Madison, created the first ES-cell line using donated (unused) embryos from a fertility clinic. The study sent a shockwave through the scientific community and general public. We now had the technology to grow human pluripotent ES cells—with the potential to develop into every cell type in the human body—in a dish! Thomson quickly became a celebrity scientist. (Thomson’s headshot was on the cover of the August 20, 2001 issue of Time Magazine, next to big text that read: “The Man Who Brought You Stem Cells”.)
However, not all were excited about the news. Backlash from conservative communities, who opposed the use of human embryos, resulted in a temporary ban on developing new ES cell lines with government funding. Nonetheless, the ban did not deter researchers from studying ES cells using private or state funding. By 2001, human ES cells have been successfully derived into neural, cardiac, hematopoietic, endothelial, and insulin-producing cells. In 2010, the first in-human clinical trial was initiated; which used human ES cell-derived materials to treat spinal cord injury.
2006 marked another milestone in stem cell research: the discovery of induced pluripotent stem (iPS) cells. Dr. Shinya Yamanaka at Kyoto University successfully reprogrammed adult fibroblasts (common cells in connective tissue that form the extracellular matrix and collagen) to revert back into an embryonic-like pluripotent state—simply by expressing four specific genes. He named these reprogrammed cells “induced pluripotent stem cells” or iPS cells. A year later, human iPS cells were made in a similar fashion by both Thomson and Yamanaka. Yamanaka later received the 2012 Nobel Prize (some argue that Thomson deserved to share the prize).
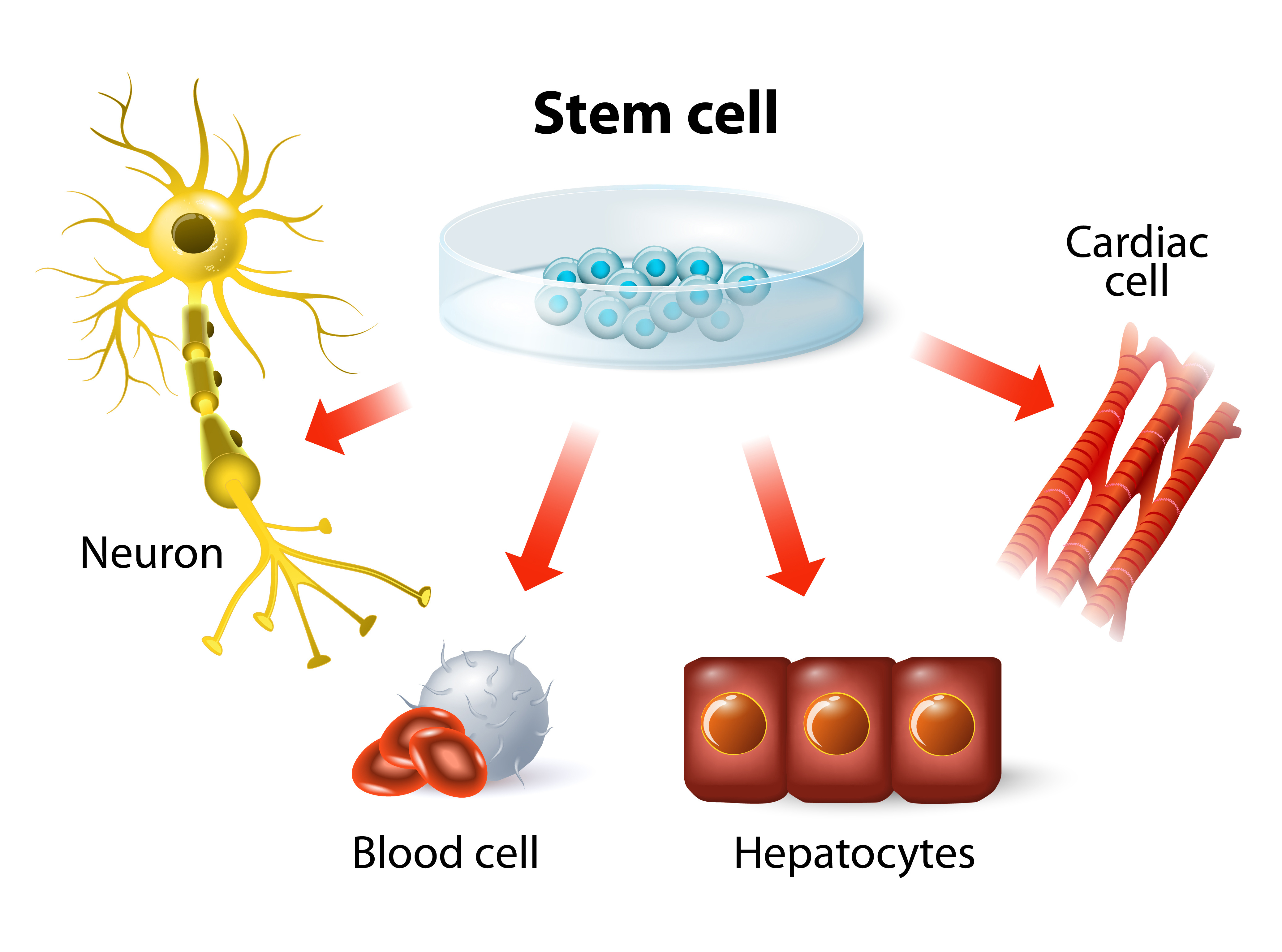
The ability to reprogram adult cells back into a pluripotent state suggested we could create an unlimited supply of pluripotent cells that genetically matched a specific individual—without the ethical baggage of using human embryos. This meant, in theory, you could take fibroblasts from a patient with a neurological disorder, such as Parkinson’s disease, revert the fibroblasts into iPS cells, edit the “faulty genes” in those cells, then redifferentiate the healthy iPS cells into neural stem cells that can be introduced back into the same patient to produce healthy neurons. Of course, this is easier said than done. The technical difficulties and high cost of generating and editing iPS cells from individual patients have complicated the development of iPS-based treatments. Currently, there is only one human clinical trial using cells derived from iPS cells, which treats macular degeneration (an incurable eye disease that leads to blindness).
Despite the emergence of iPS cells, ES cells have continued to dominate in the clinical realm. To this date, there are 18 clinical trials using ES cells to treat various disorders, including macular degeneration, Parkinson’s disease, spinal cord injury, heart disease and diabetes. The future is bright, but there is still one major problem in ES cell-based therapies. Because ES cell treatments use donor cells from other healthy individuals—not the patients’ own cells—there is a high risk of immune rejection. But no fear, scientists have a plan.
In 2017, Dr. David Russell (mentioned in the beginning of this blog) re-engineered human embryonic stem cells to remove specific proteins—human leukocyte antigens (HLA)—from the cell surface. HLA proteins allow the immune system to determine whether the presenting cell is “self” or “foreign”. Removing HLA proteins is like wrapping the foreign cell with an invisible cloak, rendering it unnoticeable by the immune system. In his talk at the Stem Cell Symposium, Russell discussed the many advantages of using these “universal donor cells (UDCs)” to treat diseases. Only one cell line is needed, which reduces the cost, complexity and time required for clinical trials. Also, it does not require immunosuppression, which weakens the patient’s immune system. Russell and many others believe that UDCs are the future of regenerative medicine. In fact, UDC-based therapies to treat cancer, macular degeneration, skin wounds and type 1 diabetes are already being developed.
It is amazing to see how far we have come over the last 20 years. Thanks to visionary scientists like James Thomson, Shinya Yamanaka, David Russell—and countless other principal investigators, post-docs and grad students who work tirelessly in the lab every day—treatments for many life-threatening diseases may be available in the near future. Nonetheless, there is still much more to learn and many more challenges to overcome. Who knows where the next 20 years will take us?

