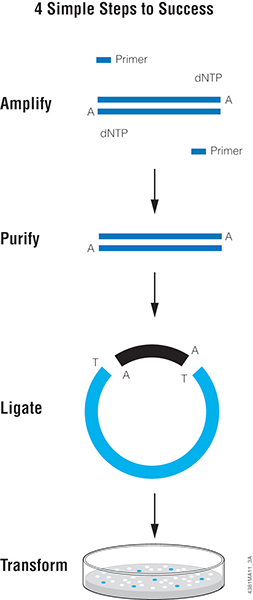
One of the easiest methods for cloning blunt-ended DNA fragments including PCR products is T-vector cloning, such as with pGEM®-T or pGEM®-T Easy Vector Systems. This method takes advantage of the “A” overhang added by a PCR enzyme like Taq DNA Polymerase. T vectors are linearized plasmids that have been treated to add 3′ T overhangs to match the A overhangs of the insert. The insert is directly ligated to the T-tailed plasmid vector with T4 DNA ligase. The insert can then be easily transferred from the T vector to other plasmids using the restriction sites present in the multiple cloning region of the T vector.
Proofreading polymerases like Pfu do not add “A” overhangs so PCR products generated with these polymerases are blunt-ended. In a previous blog, we discussed a simple method for adding an A-tail to any blunt-ended DNA fragment to enable T-vector cloning. Below, we think about the next step: Ligation.
Continue reading “Cloning with pGEM®-T Vectors: Ligation”