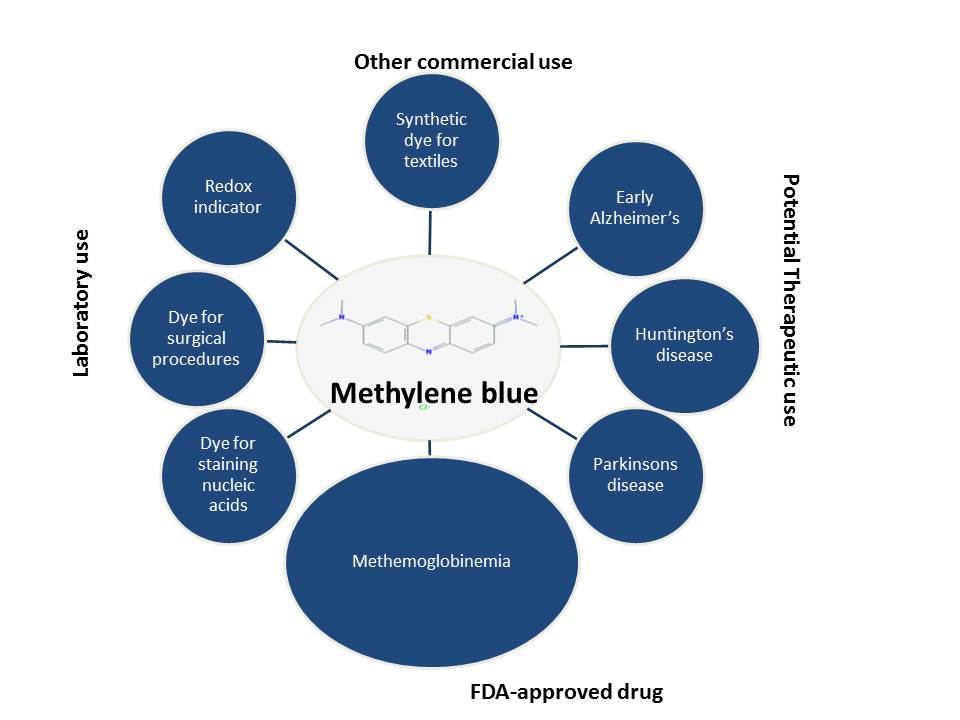
 When I first came across an article about methylene blue being protective against neuronal death, I was intrigued. I had always associated methylene blue (sometimes confused with methyl blue—an entirely different molecule) with staining nucleic acids or proteins following membrane transfer or with staining bacteria or its use as a redox indicator. It turns out that methylene blue (MB) has an extremely wide range of applications: commercial (dye in the textile / paper industry), laboratory (supravital dye, redox indicator) and an amazingly large plethora of clinical and therapeutic (early Alzheimer’s disease, mild cognitive impairment) applications(1). In fact, MB is an FDA-grandfathered antidote for the treatment of methemoglobinemia, a condition characterized by elevated levels of oxidized form of hemoglobin that interferes with its ability to release oxygen to the tissues.
When I first came across an article about methylene blue being protective against neuronal death, I was intrigued. I had always associated methylene blue (sometimes confused with methyl blue—an entirely different molecule) with staining nucleic acids or proteins following membrane transfer or with staining bacteria or its use as a redox indicator. It turns out that methylene blue (MB) has an extremely wide range of applications: commercial (dye in the textile / paper industry), laboratory (supravital dye, redox indicator) and an amazingly large plethora of clinical and therapeutic (early Alzheimer’s disease, mild cognitive impairment) applications(1). In fact, MB is an FDA-grandfathered antidote for the treatment of methemoglobinemia, a condition characterized by elevated levels of oxidized form of hemoglobin that interferes with its ability to release oxygen to the tissues.
What makes this compound, so rightly called “magic bullet” so unique and versatile? The answer partly lies in its chemical structure. One of the earliest uses of MB was as a redox indicator. In its oxidized form it accepts electrons from electron donors turning blue. In its reduced colorless form, it acts as an electron donor and can transfer electrons to oxygen to form water. Also, the unique groups in the molecule structure allow MB to auto-oxidize and allow for retention of its redox activity despite being reduced. Also MB is highly soluble in aqueous medi,a and the aromatic nature confers lipophilicity, enabling MB to penetrate cell membranes. The icing on the cake is its ability to cross the blood-brain barrier. So, the resultant molecule is a powerful antioxidant that can be targeted to the CNS.
What are the cellular effects of MB? It has affinity for various oxidases in the tissue, most of which reside in the mitochondria. It increases oxygen consumption of living cells. Not surprisingly, MB accumulates in the mitochondria—not as a passive dye, but performs the function of a natural mitochondrial electron carrier by transferring electrons to oxygen or other electron acceptors. At low concentrations, MB exerts antioxidant and electron shuttling functions, increasing cellular respiration; however at higher concentrations, it steals electrons away from the electron transport chain complex, decreasing cellular respiration. In low doses MB has been shown to decrease the production of reactive oxygen species, inhibit rotenone-induced lipid peroxidation and prevent cell death due to oxidative stress. As it is widely known, impaired mitochondrial metabolism is associated with loss of viability and neurodegeneration, which is also the hallmark of Alzheimer disease and other related disorders. Therefore, it follows that any compound capable of improving mitochondrial metabolism in the CNS would be a good candidate for a therapeutic drug. Not surprisingly, MB has been reported to be effective in treatment of mild to moderate Alzheimer disease in improving cognitive function.
Apart from its effect on mitochondrial metabolism, MB is also postulated to play a role as a modulator of aggregation thereby underlining its potential role in AD and other dementias. Huntington disease (HD) is characterized by increased polyglutamine repeat (CAG repeat at the gene level) in Huntington protein (Htt) leading to protein misfolding and aggregation. Recently, Sontag et al (2) reported the modulatory effect of MB in Huntington protein aggregation intermediates, both in in vitro and in vivo Drosophila and mouse HD models. Oral administration of MB in R6/2 mice (HD model with increased CAG repeats) delayed progression of phenotypic effects such as progressive loss of body weight and motor coordination, generally associated with HD. In addition to modulating aggregation of Htt protein, MB also reduced neurotoxicity in primary neurons.
But what about toxicity? Reports suggest that the pharmacokinetics of MB is not defined by regular drug-receptor interactions. Instead, it displays what is known as hormetic effects- that is a dose response with opposite effects at low and high doses. Higher concentrations of MB promote dimerization thereby regulating the amount of available binding sites. This theory is supported by the report that low, but not high concentrations of MB, are protective in neuronal cultures. MB is currently in clinical trials, and no visible toxicity is reported.
Therefore, the increased bioavailability of a nontoxic, CNS active compound such as MB, could, in low doses, prove to be a promising therapeutic agent for preventing various neurological and biochemical effects of diseases resulting from oxidative stress. That is a long journey for a compound originally identified as a dye.
References
- Rojas JC, Bruchey AK, Gonzalez-Lima F. (2012) Neurometabolic mechanisms for memory enhancement and neuroprotection of methylene blue. Prog Neurobiol. 96, 32-45.
- Sontag EM, Lotz GP, Agrawal N, Tran A, Aron R, Yang G, Necula M, Lau A,Finkbeiner S, Glabe C, Marsh JL, Muchowski PJ, Thompson LM. (2012) Methylene Blue Modulates Huntingtin Aggregation Intermediates and Is Protective in Huntington’s Disease Models. J Neurosci. 32,11109–19.
Anupama Gopalakrishnan
Latest posts by Anupama Gopalakrishnan (see all)
- Mitochondrial DNA Typing in Forensics - February 25, 2015
- PowerQuant System: Tool for informed casework sample processing decisions - July 21, 2014
- Biology of Overeating and the Weight-Gain Cycle - April 28, 2014

Dear Anupama,
Instead of using pure Methylene Blue ( Zinc Free ) , one should reduce Methylene Blue into it Leuco-form by addind Sodium Ascorbate and then use this Super-anti-oxidant for neuro-generative deseases.
with thanks
Dr Ashok Ghosh
dr_a_gh@yahoo.com
Thank you for your comment Dr. Ghosh. This is very interesting. I assume the leuco form is also preferred because it renders the drug more bioavailable and less toxic at higher doses?
Anti-aging activity of methylene blue.
“Methylene blue delays cellular senescence and enhances key mitochondrial biochemical pathways”
Atamna, H., et al. FASEB, 2008
For Interest in collaboration:
hatamna@tcmedc.org
Hani.atamna@CNUCOM.org