As part of my job I occasionally search the literature for papers citing use of Promega products in new or interesting ways. Any search on dual-luciferase reporters usually generates a lot of returns. A search for dual-luciferase on Highwire press generates over 700 articles from 2009 alone. So why are dual-luciferase reporter assays so widely used?
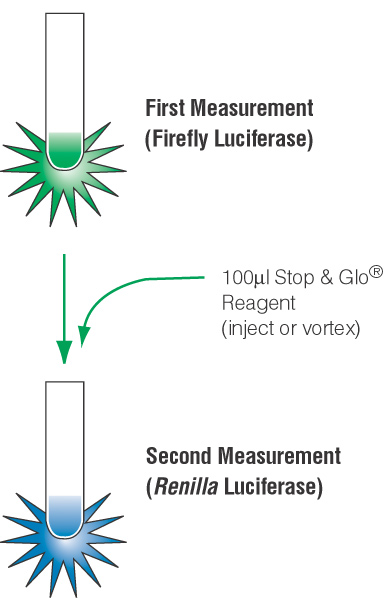 The article Normalizing Genetic Reporter Assays: Approaches and Considerations for Increasing Consistency and Statistical Significance provides a good overview of why dual-reporter systems are such a useful choice. The term “dual reporter” refers to the simultaneous expression and measurement of two individual reporter enzymes (an “experimental” and a “control” reporter) within a single system. Typically, the experimental reporter reflects the effect of the test conditions on the level of gene expression. A second reporter is used to provide an internal control to which the activity of the experimental reporter is normalized. In the dual-luciferase reporter assays, the activity of an experimental firefly luciferase reporter is compared to a control Renilla luciferase activity.
The article Normalizing Genetic Reporter Assays: Approaches and Considerations for Increasing Consistency and Statistical Significance provides a good overview of why dual-reporter systems are such a useful choice. The term “dual reporter” refers to the simultaneous expression and measurement of two individual reporter enzymes (an “experimental” and a “control” reporter) within a single system. Typically, the experimental reporter reflects the effect of the test conditions on the level of gene expression. A second reporter is used to provide an internal control to which the activity of the experimental reporter is normalized. In the dual-luciferase reporter assays, the activity of an experimental firefly luciferase reporter is compared to a control Renilla luciferase activity.
Reporter data are normalized to correct for the effect of variables such as cell number and transfection efficiency, which can have an unwanted effect on the magnitude of reporter expression. To do this, the reporter activity in a particular sample is divided by a second value specific to the same sample. The article explains why use of vector-based control reporters has advantages over other commonly used methods such as normalization based on total protein concentration or total ATP content. Normalizing the activity of the experimental reporter to the activity of the internal control reporter eliminates inherent assay-to-assay variability, such as differences in the number and health of cultured cells and in the efficiencies of transfection and cell lysis, which can undermine experimental accuracy. The article provides basic details on common methods of normalization, example data, and useful tips for interpreting results.
Read the article here to get all the details.
Isobel Maciver
Latest posts by Isobel Maciver (see all)
- 3D Cell Culture Models: Challenges for Cell-Based Assays - August 12, 2021
- Measuring Changing Metabolism in Cancer Cells - May 4, 2021
- A Quick Method for A Tailing PCR Products - July 8, 2019
