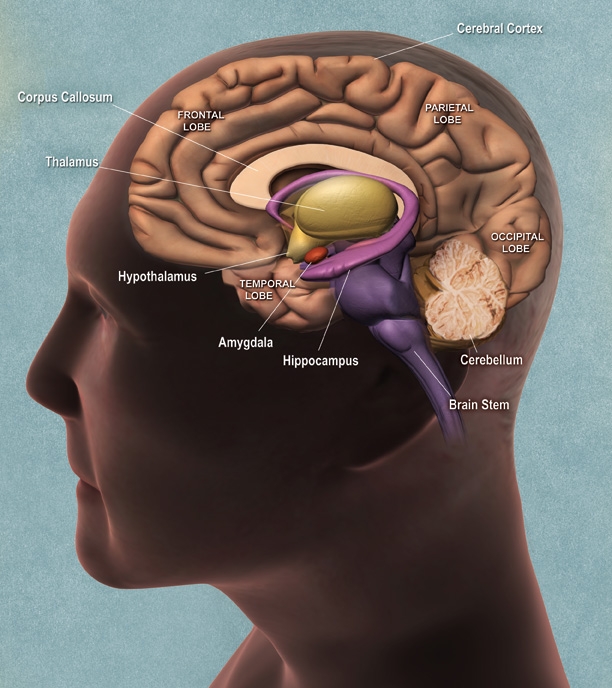
Neurogenesis (the creation of new neurons) is widely accepted as fact for the peripheral nervous system; for example, the nerves in my finger grew back even though I had lost them to frostbite. However, there is the widespread belief that neurons in the central nervous system (CNS) are static and do not multiply after adulthood. The evidence for such belief is everywhere, from spinal cord injuries that result in permanent lifelong paralysis to neurodegenerative conditions such as multiple sclerosis. However, like many widespread beliefs that are simply assumed to be true, nature always finds a way to foil our best laid plans.
As early as 1962, Joseph Altman reported that the brain could produce new neurons, an idea that flew in the face of current neurological dogma and was largely ignored (1). But Altman’s research was corroborated in the 1970’s via Michael Kaplan’s work on adult rats (2). Kaplan’s work was heavily ridiculed, eventually ending his career as a research scientist.
And yet, the evidence continued to build. By the 1990’s, the scientific community finally accepted that neurogenesis is indeed a critical facet of the CNS, especially in the brain. And as soon as the old dogma was shaken off, enthusiasm and interest started to peak on another front: what could we do to stimulate neurogenesis within ourselves? More than anything else, what could we do to encourage neurogenesis in our brains in order to (hopefully) stave off such frightening diseases as Alzheimer’s and Parkinson’s?
Look up the words “brain training” via Google and you will be buried in a slew of software programs, courses, vitamins, and even iPhone apps all claiming to improve your memory and cognitive function. However, there is very little evidence that these programs work and no clear mechanism outlined on how “new and improved” neurons are generated. Even those traditionally recommended brain exercises such as doing crossword puzzles and word teasers may only serve to make you really good at…doing crossword puzzles and word teasers. Meanwhile, you’ll still be struggling to remember where you left your car keys.
As far as nutrition is concerned, the NIH recently conducted an evaluation of many purported cognitive enhancers such as vitamin B6, B12, folic acid, flavenoids, alcohol and omega-3 and found the actual evidence to be weak (3). Even the widely advertised Mediterranean diet, when scrutinized for actual cognitive enhancement, was found to work for only a small subset of individuals. These data are not surprising given the fact that positive correlation studies are often lauded by journalists and scientists alike, while null studies are frequently buried or ignored.
So, is there a tried and true method for inducing neurogenesis? Yes. However, this method involves significantly more work than just popping a pill or eating the right foods.
When Charles Hillman of the University of Illinois measured the body mass index of 259 3rd and 5th graders and then put all grade-schoolers through some exercise routines, he found that the kids who were more fit performed better on subsequent math and verbal tests (4). Hillman’s research was followed by a more recent study wherein study volunteers engaged in a three-month fitness program and then had their brain cell density measured. The volunteers that had exercised for only three months showed significantly more brain cells than when they had just started their exercise regimen. Such findings fall in line with what other researchers have also discovered, such as the fact that exercise even in older adults can result in denser interconnections between brain cells.
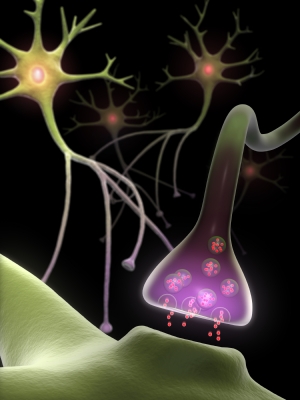
While increased blood flow and oxygen may partly explain why exercise results in neurogenesis, the molecular reason is far more complex. During contraction, muscle tissues secrete a compound called IGF-I (insulin-like growth factor I). When IGF-I reaches the brain, it induces the release of another compound termed BDNF (brain-derived neurotrophic factor). BDNF is like brain fertilizer, inducing nerve cells to grow and connect with one another. Such branching neurons form the basis of learning, memory and reasoning.
Even more stunning is what happens when a brain experiences a low level of BDNF. At the UCLA, Fernando Gómez-Pinilla had two groups of rats perform wheel-running exercise routines for two weeks (5). He then took these animals and injected half of them with a BDNF-blocking chemical. The entire rat group was later challenged with locating an underwater object. The no-BDNF blocker group was able to find the submerged item easily; the BDNF blocker group was not so fortunate.
The growth factor BDNF is produced at a steady level throughout adulthood but does start dwindling in old age. This is partly why it is harder for older adults to learn new things, retain memories and, quite often, form sound judgments. Because it was previously assumed that neurogenesis did not happen past childhood, researchers figured forgetfulness and dementia to be the end products of aging. However, thanks to recently accumulated evidence, this theory is no longer true.
But what about the effects of exercise on older adults who are already at risk of or starting to lose cognitive function? Arthur Kramer of the University of Illinois analyzed men and women in their sixties and seventies who had engaged for several weeks in regular aerobic exercise (7). He then used MRI (magnetic resonance imaging) to study their various brain regions. What Arthur discovered was that older adults who had engaged in aerobic exercise showed an actual increase in the size of their frontal brain lobes. In addition, immediately upon ceasing aerobic activity, the older adults could better and more accurately answer learning-based questions. What Kramer concluded from his studies was that not only can exercise increase brain size over the long-term, but in the short-term it also causes the immediate production of learning and memory-associated neurotransmitters and endorphins like dopamine, serotonin and norepinephrine.
Unfortunately, when it comes to neurotransmitters, endorphins and neural growth, the brain is always operating on a “use it or lose it” mechanism. After only a month of inactivity, astrocytes begin to shrink and neurons don’t function as well as they used to, according to William Greenough, who is a psychologist at the University of Illinois (4). Thus, continued and lifelong exercise is the only way in which one can continue to stay sharp.
Exactly how much exercise is required before neurogenesis and a decreased risk of dementia and memory loss result? The U.S. Centers for Disease Control and Prevention recommend that you exercise 30 minutes for 5 days a week if the exercise is of moderate intensity (i.e. if you can perform the exercise while holding a conversation). Alternately, you can exercise 20 minutes for 3 days a week if the exercise is of high intensity (i.e., if you are unable to perform the exercise and hold a conversation).
- ALTMAN J (1962). Are new neurons formed in the brains of adult mammals? Science (New York, N.Y.), 135, 1127-8 PMID: 13860748
- Kaplan MS (2001). Environment complexity stimulates visual cortex neurogenesis: death of a dogma and a research career. Trends in neurosciences, 24 (10), 617-20 PMID: 11576677
- Plassman BL, Williams JW Jr, Burke JR, Holsinger T, & Benjamin S (2010). Systematic review: factors associated with risk for and possible prevention of cognitive decline in later life. Annals of internal medicine, 153 (3), 182-93 PMID: 20547887
- Can Exercise Make you Smarter?
- Molteni R, Ying Z, & Gómez-Pinilla F (2002). Differential effects of acute and chronic exercise on plasticity-related genes in the rat hippocampus revealed by microarray. The European journal of neuroscience, 16 (6), 1107-16 PMID: 12383240
- Pereira AC, Huddleston DE, Brickman AM, Sosunov AA, Hen R, McKhann GM, Sloan R, Gage FH, Brown TR, & Small SA (2007). An in vivo correlate of exercise-induced neurogenesis in the adult dentate gyrus. Proceedings of the National Academy of Sciences of the United States of America, 104 (13), 5638-43 PMID: 17374720
- Erickson KI, & Kramer AF (2009). Aerobic exercise effects on cognitive and neural plasticity in older adults. British journal of sports medicine, 43 (1), 22-4 PMID: 18927158
- How much physical activity do adults need?
Halina Zakowicz
Latest posts by Halina Zakowicz (see all)
- 10 Surprising Facts About Hallucinogens, Psychedelics and “Magic Mushrooms” - May 11, 2011
- How Exercise Can Grow Your Brain - April 11, 2011

Fascinating stuff! It is great to see how our body works in a coordinated fashion for its benefit. The chemical pathways are so intricate. I knew that exercise could help with mental conditions – I guess there is a link with what is said in this blog. If only people would take responsibility for their fitness by looking around for things like best Elliptical fitness machines where they can exercise in the privacy of their own homes